
In evaluating a child with suspected obstructive sleep apnea (OSA), it is important to look for evidence of crossbite and for abnormalities of the hard palate that may be amenable to treatment by maxillary expansion, thus avoiding the need for surgery and/or chronic use of positive airway pressure. The case of a 13-year-old obese girl with moderate OSA demonstrates the success of maxillary expansion in bringing about resolution of the moderate obstructive sleep apnea despite the patient’s continued weight gain.
PATIENT CASE
A 13-year-old obese girl was referred for a consultation to the sleep disorders clinic because of findings of moderate obstructive sleep apnea seen on a sleep study done 8 months prior to the visit. On that study, 51 hypopneas were scored, giving an apnea-hypopnea index of 7/hour. The obstructive events were more prominent while she was in the supine position, lasted 8-17 seconds, and were associated with desaturations into the mid 90% range.
The patient presented to the clinic accompanied by her mother, who reported that her daughter had “always snored loudly,” that there had been no change in her snoring since the study, and that it was easily audible outside her bedroom. Her mother also reported that she had observed occasional apneic episodes lasting “a couple of seconds,” and that although she had never actually seen or heard her daughter choke or gasp for air, she was certain that she was having trouble breathing while asleep. The child herself denied night sweats, nocturnal enuresis, restless sleep, breathing through an open mouth, or awakening with a dry mouth on a regular basis. She also denied excessive daytime sleepiness, but stated that she would fall asleep and/or nap during the day if she had not gotten sufficient sleep the night before, and that this was happening once every week or two.
The patient’s schedule was as follows: she would typically go to bed at 9:30 pm, fall asleep by 10:15 pm, and awaken at 6:30 am in the morning, thus sleeping about eight and a quarter hours every night. On Saturday mornings, she was generally out of bed by 7:30 am so she could attend synagogue services. On Saturday nights, she would generally stay up later than on other nights and would sleep in on Sundays, usually getting out of bed between 9:30 am and 10 am.
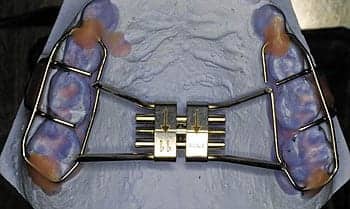
The review of systems was notable for the following: she was obese and had recently started seeing a nutritionist to help bring her weight under control. Respiratory: she had mild asthma for which she was being treated with budesonide as a controller medication. Orthodontics: she had braces as well as a palatal expander placed 2 months earlier to correct three discrete areas of crossbite. While the palatal expander was supposed to remain in place for three additional months, she explained that, as she had found that it was pressing down upon her tongue and causing her discomfort, she was hoping to have it removed the following week. The remainder of the extensive systems review was otherwise unremarkable.
The patient’s medical history was notable for removal of her adenoids in the first year of life. Her tonsils were intact.
Medications at the time of the clinic visit included budesonide, albuterol, and lansoprazole.
The family history was notable for her father and maternal aunt having obstructive sleep apnea, and for diabetes mellitus type 2 in the family.
PHYSICAL EXAM
Upon examination, the patient was a pleasant obese girl with no signs of distress. Her color was good with no cyanosis, clubbing, or edema of the extremities. Her height was 171.4 cm, weight 88 kg, body mass index 30 kg/m2. Her blood pressure was 103/66 and her pulse 64. Her tympanic membranes were normal in appearance, her pupils equal and responsive to light and accommodation. Her nasal septum appeared mildly deviated toward the right, and she had more difficulty breathing in through her right nostril than her left. Her palate was Mallampati I, and she had +1 tonsils. While no obvious crossbite was apparent, she did have braces and a palatal expander, which left a visible indentation on her tongue and which clearly prevented her tongue from abutting the hard palate. She was noted to have mild retrognathia. Her neck was supple without lymphadenopathy, its circumference was 38 cm, and acanthosis nigricans was noted. Her breath sounds were clear and equal bilaterally without crackles or wheezes. Her heart sounds were normal S1, S2 without murmurs. Her abdomen was soft, supple, nontender, and nondistended with no organomegaly.
ASSESSMENT
- Moderate obstructive sleep apnea
- Obesity
- Narrow palate undergoing expansion
- Mild retrognathia
- Excessive daytime sleepiness
- Insufficient sleep
INTERVENTIONS AND RECOMMENDATIONS
Weight loss as a long-term strategy was encouraged, though despite her work with the nutritionist, her weight had decreased by only 200 grams during the 8 months between the original sleep study and the clinic appointment. Because of her age, obesity, and small size of her tonsils, it seemed unlikely that tonsillectomy—either alone or in combination with revision adenoidectomy (if there had been regrowth)—would effectively cure the obstructive sleep apnea. The use of continuous positive airway pressure was discussed with the patient and her mother.
It was also pointed out that it was possible that persevering with the maxillary expansion might prove sufficient in opening up enough room in her upper airway and preventing the retrusion of her tongue to relieve the obstruction seen on the sleep study, and avoid the need for further interventions such as surgery or continuous positive airway pressure. The patient and her mother agreed to keep the maxillary expander in place for the period originally recommended by the orthodontist, and to await the results of a follow-up sleep study, which was scheduled for 1 week after its removal. Counseling was also given about sleep hygiene, the importance of maintaining a regular sleep schedule, and age-appropriate sleep requirements.
FOLLOW-UP
After the maxillary expander was removed, the follow-up sleep study was done as planned, and showed mostly quiet breathing with very infrequent snoring, an absence of obstructive or central events, and normal gas exchange and heart rate. These findings were all the more impressive in light of the fact that the patient had continued to gain weight, and on the night of the study weighed 91.3 kg and had a body mass index of 30.6 kg/m2.
Upon examination subsequent to the study, she no longer had any evidence of crossbite. At the follow-up clinic visit after the sleep study, the patient reported an improvement in her daytime sleepiness, and her mother no longer expressed any concerns about her breathing during sleep. The patient was advised to continue to try to lose weight, and to follow up should there be a change in her breathing patterns going forward.
DISCUSSION
Obstructive sleep apnea is present in 1% to 4% of the general pediatric population,1 with a higher incidence in certain populations such as children with Down syndrome2,3 and Prader-Willi syndrome.4 The incidence of obstructive sleep apnea peaks between the ages of 3 and 6,5 which is when the tonsils and adenoids are largest in relation to the dimensions of the upper airway.
In the general population, obstructive sleep apnea is known to cause excessive daytime sleepiness, hypertension,6 poor glucose metabolism,7 and increased cardiovascular8 and cerebrovascular disease.9 In addition, children with obstructive sleep apnea can suffer from failure to thrive,10 and from behavioral, cognitive, and developmental impairment. Many studies have found a clear connection between obstructive sleep apnea and a reduction in verbal IQ,11 decreased executive function,12 lower Bailey developmental scores,13 poor school performance,14 and attention deficit and hyperactivity disorder.15
The traditional interventions for obstructive sleep apnea in children usually include the medical and/or surgical treatment of adenotonsillar hypertrophy, and positive airway pressure. However, it is important to remember that maxillary expansion is also an effective way of treating obstructive sleep apnea in children and adolescents.16-18
Dennis Rosen, MD, is a pediatric pulmonologist and sleep specialist at Children’s Hospital Boston, where he is a member of the Division of Respiratory Diseases and associate medical director of the Sleep Disorders Program. He is also an instructor in pediatrics at Harvard Medical School. The author can be reached at [email protected].
REFERENCES
- Lumeng JC, Chervin RD. Epidemiology of pediatric obstructive sleep apnea. Proc Am Thorac Soc. 2008;5:242-252.
- Stebbens VA, Dennis J, Samuels MP, et al. Sleep related upper airway obstruction in a cohort with Down’s syndrome. Arch Dis Child. 1991;66:1333-1338.
- Marcus CL, Keens TG, Bautista DB, et al. Obstructive sleep apnea in children with Down syndrome. Pediatrics. 1991;88(1):132-139.
- O’Donoghue FJ, Camfferman D, Kennedy JD, et al. Sleep-disordered breathing in Prader-Willi syndrome and its association with neurobehavioral abnormalities. J Pediatr. 2005;147(6):823-829.
- Rosen CL. Obstructive sleep apnea syndrome (OSAS) in children: diagnostic challenges. Sleep. 1996;19(10 suppl):S274-277.
- Li AM, Au CT, Sung RYT, et al. Ambulatory blood pressure in children with obstructive sleep apnoea—a community based study. Thorax. 2008;63:803-809.
- Tamura A, Kawano Y, Watanabe T, Kadota J. Relationship between the severity of obstructive sleep apnea and impaired glucose metabolism in patients with obstructive sleep apnea. Respir Med. 2008;102:1412-1416.
- Parish JM, Somers VK. Obstructive sleep apnea and cardiovascular disease. Mayo Clin Proc. 2004;79(8):1036-1046.
- Nishibayashi M, Miyamoto M, Miyamoto T, Suzuki K, Hirata K. Correlation between severity of obstructive sleep apnea and prevalence of silent cerebrovascular lesions. J Clin Sleep Med. 2008;15;4(3):242-247.
- Freezer NJ, Bucens IK, Robertson CF. Obstructive sleep apnoea presenting as failure to thrive in infancy. J Paediatr Child Health. 1995;31(3):172-175.
- Halbower AC, Degaonkar M, Barker PB, et al. Childhood obstructive sleep apnea associates with neuropsychological deficits and neuronal brain injury. PLoS Med. 2006;3(8):e301.
- Karpinski AC, Scullin MH, Montgomery-Downs HE. Risk for sleep disordered breathing and executive function in preschoolers. Sleep Med. 2008;9:418.
- Montgomery-Downs HE, Gozal D. Snore-associated sleep fragmentation in infancy: mental development effects and contribution of secondhand cigarette smoke exposure. Pediatrics. 2006;117:e496-e502.
- Gozal D. Sleep-disordered breathing and school performance in children. Pediatrics. 1998;102:616-620.
- Chervin RD, Ruzicka DL, Giordani BJ, et al. Sleep-disordered breathing, behavior, and cognition in children before and after adenotonsillectomy. Pediatrics. 2006; 117:e769-e778.
- Villa MP, Malagola C, Pagani J, et al. Rapid maxillary expansion in children with obstructive sleep apnea syndrome: 12-month follow-up. Sleep Med. 2007;8(2):128-134.
- Pirelli P, Saponara M, Guilleminault C. Rapid maxillary expansion in children with obstructive sleep apnea syndrome. Sleep. 2004;27:761-766.
- Cistulli PA, Palmisano RG, Poole MD. Treatment of obstructive sleep apnea syndrome by rapid maxillary expansion. Sleep. 1998;21:831-835.






