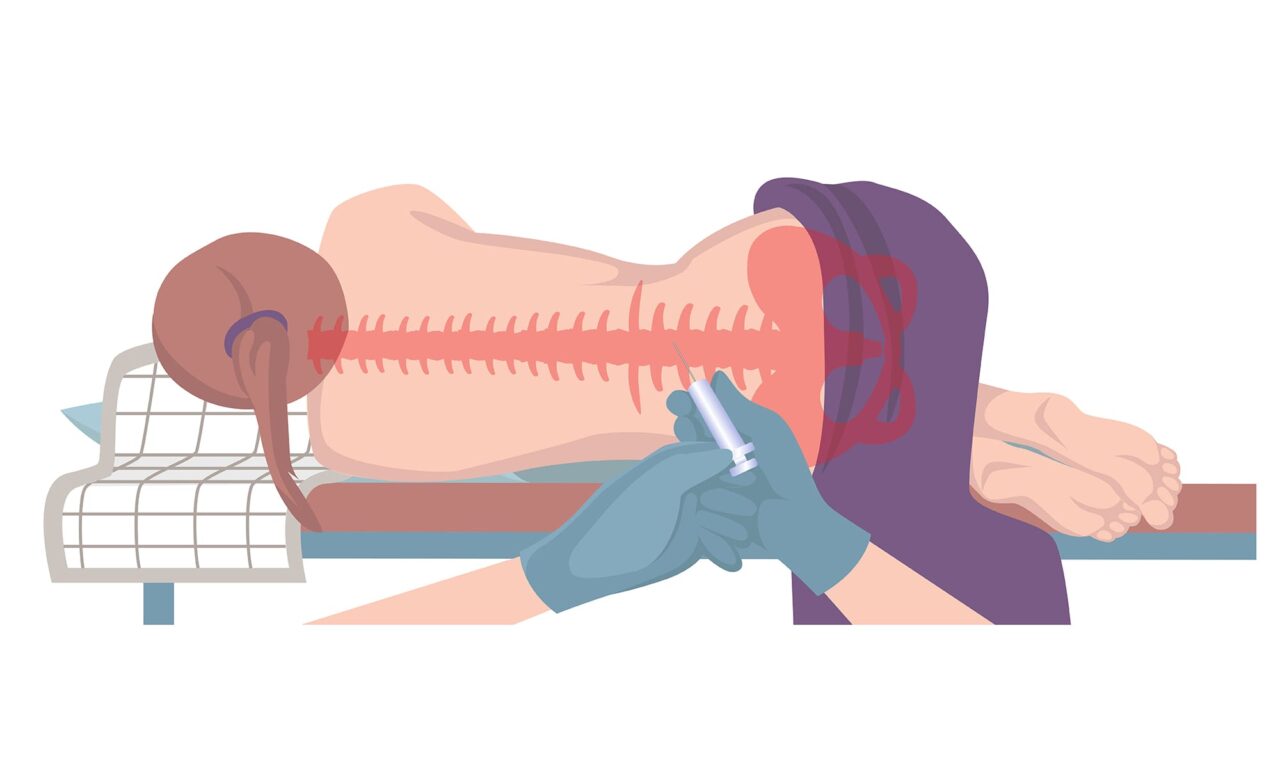
How Orexin-A Testing Could Become Easier
The gold-standard assay for measuring orexin deficiency in cerebrospinal fluid requires a lumbar puncture and radioactive materials. New findings could help advance testing alternatives for people with narcolepsy.