 |
As the recession lingers, more and more people are losing their jobs and therefore are likely to have no insurance coverage for health care. With increasing loss of health insurance coverage, there is an overwhelming need for improvisation and compassion. JC, a truck driver without health insurance coverage, was on the verge of losing his job due to obstructive sleep apnea-hypopnea syndrome (OSAHS). Through insight into these matters, the Southwest Cleveland Sleep Center was able to arrange a payment plan for not only his diagnosis with overnight polysomnography but follow-up with a sleep specialist and long-term treatment. Such flexibility is the need of the hour and can have a lasting impact on personal lives and the community at large.
BACKGROUND
OSAHS is a common disorder.1 It is characterized by upper airway collapse and results in sleep fragmentation, excessive daytime sleepiness, reduced quality of life,2 and increased rate of motor vehicle accidents (MVA).3 A patient with OSAHS is seeking help for one of the basic necessities of life—a good night’s sleep; and a sleep medicine professional with a managed care plan can be literally a lifesaver.
The economic burden related to untreated OSAHS adds up to billions of dollars per year.4 It has been identified as an independent risk factor for long-term sick leave and permanent disability.5 Studies have also shown that CPAP is an effective and cost-efficient treatment for OSAHS compared with conservative measures and placebo in populations with moderate to severe daytime sleepiness.6
|
Subscribe to Sleep Report to receive updates on OSA and drowsy driving. |
Untreated OSAHS is not only costly but dangerous for patients and, potentially, the community at large. For the OSAHS patient population that works in certain industries such as construction or those that involve operating heavy machinery or driving, the screening, diagnosis, and treatment of OSAHS are particularly significant.
OSAHS among commercial drivers increases the risk of fall-asleep crashes, which incur large expenses. It has been strongly suggested that screening with BMI, age, and gender, followed by confirmatory in-lab polysomnography only on high-risk drivers, is cost-effective, as long as a high proportion (73.8%) of screened drivers accepts treatment.7 With an incremental cost-effective ratio (ICER) of $2,979 per quality-adjusted life year, using the Markov decision analytical model, CPAP therapy for OSAHS patients is a highly efficient use of health care resources.8
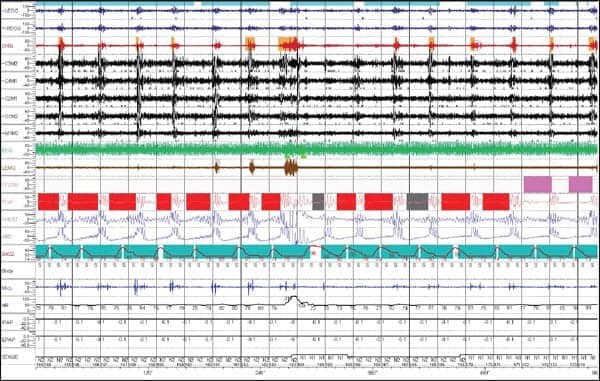 |
| Figure 1. Excerpt from overnight PSG study graph (summary). |
PATIENT CASE
JC, a 50-year-old truck driver, was referred to our clinic by a local primary care physician. JC did not have health insurance coverage, so he was considered a candidate for the Southwest Sleep Center’s pro bono, community-based program to treat underserved sleep disorder patients. To determine eligibility, comprehensive information, including insurance data, was faxed from the referring physician’s office; however, at the onset of receipt, the focus is more on the pathway for clinical care and severity of symptoms and then insurance. All patients are booked for the appropriate test—insured and uninsured. For patients without insurance and with a verified economic situation using general established poverty guidelines, dependents, and other aspects of social history (job loss, etc), a telephone interview and evaluation is conducted by a designated employee who is experienced with insurance policy. This information is combined and verified from information presented by the referring primary care physician’s office.
JC presented to our clinic with symptoms of loud snoring, witnessed apneas during sleep, leg movement during sleep, and daytime sleepiness for a long period of time. He was going to bed at 11 to 12 pm almost every night and waking up at 7 to 8 am. He reported that he has been told that he snores really loudly and even stops breathing occasionally during the night. He reported four or five episodes of nocturnal awakenings, usually gasping for breath or to void, but he was able to fall back asleep fairly quickly. Despite these episodes, he was getting an average sleep of 7 to 8 hours per night. He also complained of having night sweats and restless/disturbed sleep.
JC also reported difficulty waking up easily in the morning and had to use a loud alarm clock. He complained of waking up fatigued, having headaches after he woke up, feeling sleepy throughout the day, and easily falling asleep at inappropriate occasions. He also reported falling asleep while driving, which was not only adversely affecting his health but severely affecting his work. He was requested to mark his responses on the Epworth Sleepiness Scale (ESS). He scored 24/24 on ESS, highly suggestive of severe daytime sleepiness.
JC also reported associated complaints of episodes of shortness of breath and nocturnal indigestion. He denied any parasomnias or any other sleep-related disorder. He denied history of hypnotic intake, cataplexy, mood disorder, or taking naps during the day. There was no family history of sleep apnea. He had hypertension, which was controlled with hydrochlorothiazide diuretic medication. On general examination, JC was overweight. His throat examination revealed a narrow oropharynx that corresponded with Mallampati classification II, and his neck circumference was 18 cm.
The patient was given sleep hygiene instructions and asked to maintain a sleep/wakefulness diary. An overnight PSG was ordered, and the patient was requested to follow up after the study.
DIAGNOSIS
JC is a 50-year-old patient with a history of snoring and daytime sleepiness. The patient’s Epworth Score was 24/24. Therefore, a complete polysomnography study was performed to rule out sleep-disordered breathing and to determine the optimal level of CPAP/bi-level airflow pressure. During the study, EEG, EOG, and EMG monitored the sleep stages. Respiratory variables included: (1) Airflow monitoring by thermistors at the nose and mouth; (2) Respiratory movements by chest wall and abdominal impedance; (3) Snoring detection by microphone; (4) CPAP pressure; (5) Arterial oxygenation monitored by a finger pulse sensor; (6) Heart monitored by chest wall leads; and (7) Leg movements recorded by anterior tibial EMG sensors.
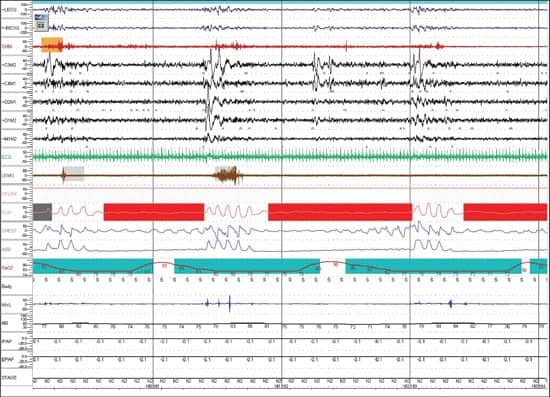 |
| Figure 2. Expanded (on time scale) excerpt from overnight PSG study graph. It clearly show apneas marked in red. As the patient stops breathing, the output from the flow sensor detects no flow and gives a flat line on the graph. The blue line below (MicL) shows snoring sound, as pulses, recorded by the microphone during the study. It is interesting to note that the oxygen saturation (SaO2) goes down during each apnea episode, as expected. |
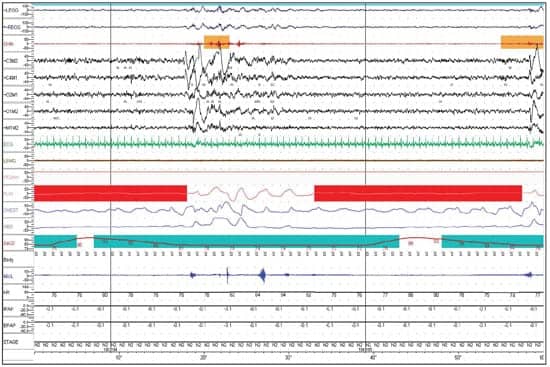 |
| Figure 3. Graph showing single apnea episode captured on PSG. It clearly shows the EEG disturbances, waking up during sleep, at the end of the apnea episode as the patient gasps for air followed by transient normal flow. |
Sleep Parameters
Total recording time was 396 minutes. Total sleep time was 336 minutes with a sleep onset of 2.5 minutes. Sleep efficiency was at 84.8%. There were three REM episodes noted totaling 75.5 minutes (22.5% of total sleep time; N = 20%). Delta sleep stage III was 12.2% and stage IV was 0.0% of total sleep time (N = 15%). EEG arousals were noted at a frequency of 20.7/h.
Cardiorespiratory Parameters
Snoring was loud. Baseline respiratory recording without CPAP for 396 minutes revealed a total of 66 obstructive, 25 mixed, and 14 central apneas and 50 hypopneas. During this period, the respiratory disturbance index (RDI) was 85/h. The mean duration of the apnea/hypopnea index was 22.2 seconds. Baseline awake arterial oxygen saturation was low at 96%. During apnea/hypopnea events, the patient’s arterial oxygenation intermittently dropped into the 92% range in REM and 90% range in NREM. The lowest arterial oxygenation was noted to be 68%; 68.0 minutes of sleep were spent at arterial saturation <90%. REM supine sleep was 8.1% associated with a worsening of respiratory events. CPAP was initiated at 4 cm and titrated up to 11 cm. The patient spent a total of 60 minutes at final CPAP level settings, which included REM sleep and supine posture. At 11 cm, the patient showed improvement. The respiratory disturbance index was reduced to 00/h. Oxygen saturations were stable at 90%.
Cardiac Rhythm
|
Sleep Stage |
Wake |
NREM |
REM |
|
Mean Heart Rate |
97.8 bpm |
79.7 |
87.6 |
Motor activity/periodic limb movements (PLMS) were noted at 20.7/h.
Clinical history and results of the sleep study confirmed the diagnosis of severe obstructive sleep apnea syndrome. The patient’s excessive daytime sleepiness is most likely resulting from OSAS. During the split night study, the patient showed excellent improvement at CPAP of 11 cm. Improved sleep architecture, including increased REM sleep (REM rebound phenomenon), reduced arousals, and decreased sleep fragmentation, suggested good therapeutic response to CPAP.
TREATMENT
After discussing the results of the sleep study and therapeutic options with the patient, he was initiated with CPAP therapy at 11 cm. The patient was encouraged to comply with therapy and have adequate sleep, ie, 8 hours per day. He was followed up with regular clinic and CPAP clinic visits. With 11 cm, he had remarkable improvement in his sleep apnea. JC used CPAP regularly, and his minor issues with mask type and comfort were resolved at clinic visits. As far as his sleep apnea symptoms were concerned, JC did very well. His Epworth Sleepiness Score improved from 24/24 to 18/24 immediately and then over time to 3/24. The patient was also encouraged to lose weight and was making some efforts in this regard. As a result of his overall improvement in sleep quality, aspects of his drowsy driving improved as manifested in his ESS score and positive episodes, which were narrated to our center sleep specialist about overall quality of life.
Prior to initial evaluation, a monthly payment plan for PSG and follow-up was also arranged for JC for $75 per month for 6 months. In this case, our in-house insurance specialist established that JC can afford overall about $450 and he agreed to make monthly installment payments. After the sleep study and a diagnosis of OSA was made, a CPAP order was prepared and then assigned to a local home care company, which will set up the patient with a refurbished CPAP unit and new accessories. In JC’s case, he received a free CPAP machine and paid the home care company for the accessories at a discounted price.
Southwest Cleveland Sleep Center works with several local home care companies in the Metro Cleveland area, including BMS Medical Supply, Apria, Lincare, and American Homepatient. As a result of assigning all of the center’s therapy orders to various home care companies, Southwest Cleveland Sleep Center has developed a good working relationship that includes combined efforts to educate the local physician community about sleep disorders through in-service and education seminars, and also the community at large through health and wellness fairs, corporate screenings, etc. This team spirit has resulted in the overall increase of screenings for sleep problems in our community. Several equipment manufacturers have been very generous to the home care companies by providing some equipment, which overall assists and facilitates these types of programs.
REFERENCES
- Young T, Palta M, Dempsey J, Skatrud J, Weber S, Badr S. The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med. 1993;328(17):1230-5.
- Flemons WW. Clinical practice. Obstructive sleep apnea. N Engl J Med. 2002;347(7):498-504.
- Teran-Santos J, Jimenez-Gomez A, Cordero-Guevara J. The association between sleep apnea and the risk of traffic accidents. Cooperative Group Burgos-Santander. N Engl J Med. 1999;340(11):847-51.
- AlGhanim N, Comondore VR, Fleetham J, Marra CA, Ayas NT. The economic impact of obstructive sleep apnea. Lung. 2008;186(1):7-12.
- Sivertsen B, Overland S, Glozier N, Bjorvatn B, Maeland JG, Mykletun A. The effect of OSAS on sick leave and work disability. Eur Respir J. 2008;32(6):1497-503.
- McDaid C, Griffin S, Weatherly H, et al. Continuous positive airway pressure devices for the treatment of obstructive sleep apnoea-hypopnoea syndrome: a systematic review and economic analysis. Health Technol Assess. 2009;13(4):iii-iv, xi-xiv, 1-119, 143-274.
- Gurubhagavatula I, Nkwuo JE, Maislin G, Pack AI. Estimated cost of crashes in commercial drivers supports screening and treatment of obstructive sleep apnea. Accid Anal Prev. 2008;40(1):104-15.
- Tan MC, Ayas NT, Mulgrew A, et al. Cost-effectiveness of continuous positive airway pressure therapy in patients with obstructive sleep apnea-hypopnea in British Columbia. Can Respir J. 2008;15(3):159-65.
Nazima K. Ahmed, MBA, is chief visionary officer at Southwest Cleveland Sleep Center (locations at Middleburg Heights, Westlake, Lutheran, Beachwood, and Parma). Fawad Taj, MD, is a research associate at Southwest Cleveland Sleep Center. Taj also recently entered the Harvard Medical School Psychiatry Residency Training Program. The authors can be reached at [email protected].






