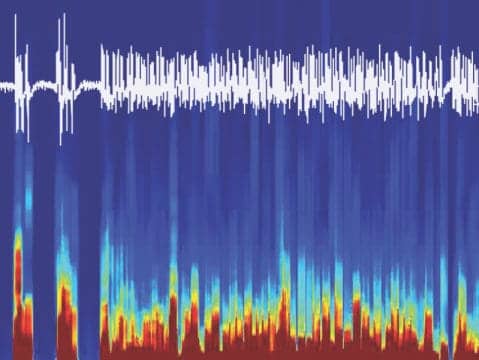
Scientists from the University of Bern have discovered a mechanism that is responsible for the rapid arousal from sleep and anesthesia in the brain. The results of their study suggest new strategies for the medical treatment of sleep disorders and recovery of consciousness in vegetative states.
“The consequences of sleep perturbations on life quality go far beyond daytime sleepiness and mood alteration. Cognitive impairment, hormonal imbalance, and high susceptibility to cardiac or metabolic disorders are amongst some of the negative impacts frequently associated with subtle chronic sleep problems,” says Antoine Adamantidis, PhD, from the Department of Clinical Research of the University of Bern and Department of Neurology at the Bern University Hospital, in a release.
The quantity and the quality of sleep are now considered as an early marker of many neurological disorders including Alzheimer’s disease, Parkinson’s disease, and schizophrenia. Unfortunately, pharmaceutical strategies combined with improved life hygiene have limited effect. “Personalized medicine” strategies for the treatment of either insufficient sleep quality or quantity are missing, the scientists say.
Brain Circuits for Arousal, Consciousness
Therefore, intensive experimental research was conducted to understand how brain circuits control sleep-wake cycle and consciousness. Together with fellow researcher Carolina Gutierrez Herrera and colleagues from Germany, Adamantidis made a dual discovery: his team identified a new circuit in the brain of mice whose activation causes rapid wakefulness while its inhibition deepens sleep. The study was published in the journal Nature Neuroscience.
Mammalian sleep is classically divided in two phases, including non-rapid eye movement (NREM) sleep and REM (or paradoxical) sleep. Key brain circuits for those two states have been identified.,However, the precise underlying mechanisms-such as the onset, maintenance, and termination of sleep and dreaming-remain unknown.
Adamantidis and Gutierrez Herrera identified a new neural circuit between two brain regions called hypothalamus and thalamus, which have been associated with electroencephalogram (EEG) rhythms during sleep. The activation of this circuit signals the termination of light sleep: using a recent technology called optogenetics, the researchers made neurons from the hypothalamus controllable with millisecond-timescale light pulses and showed that their transient activation during light sleep induced rapid awakenings, while their chronic activation maintains prolonged wakefulness. In contrast, optogenetic silencing of this circuit stabilizes light sleep and increases its intensity. In a translational analogy, hyperactivity of this circuit may cause insomnia, while its hypo-activity could be responsible for hypersomnia, making it a new therapeutical target for sleep disorders.
Causing Emergence from Anesthesia, Unconsciousness
Interestingly, the arousal power of this circuit is so strong that its activation precipitates emergence from anesthesia and the recovery of consciousness. “This is exciting discovery since therapeutical approaches to recover from a vegetative or minimally conscious state are quite limited,” says Adamantidis. Non-selective deep brain electrical stimulation has been used with some success, however the underlying brain mechanisms remain unclear. In this study, Adamantidis, Gutierrez Herrera, and collaborators nailed down a selective brain circuit important for the recovery of consciousness.
The dual findings of the Bernese researchers shine light on the brain mechanism of arousal and opens new door for tailored medical treatment of sleep perturbances, and provide a roadmap for arousing patients from a vegetative or minimally conscious state. However, Adamantidis emphasizes that “even though we made an important step forward now, it will take some time before novel therapeutical strategies will be designed based on our results.”
Image: Arousal power of the discovered circuit: Illustration of an emergence from anesthesia shown on the EEG recordings from the mouse brain controlled with optogenetics. Courtesy Department of Clinical Research, University of Bern