Data published about pediatric polysomnography can be excellent reference guidelines for scoring respiratory parameters in children and adolescents.

AMBULATORY TESTING
The overnight polysomnogram is a cumbersome and expensive test, and efforts continue to establish alternative means of diagnosing SDB. Although there has been a great deal of interest in developing home ambulatory monitoring for diagnosing sleep disorders in children, there are few studies3-5 evaluating the efficacy of home-based testing for SDB. Pulse oximetry for screening SDB has been evaluated using different devices. Brouillette et al3 compared overnight oximetry studies to complete overnight polysomnogram studies in 349 children with suspected obstructive sleep apnea (OSA). Oximetry had a positive predictive value of 97% and a negative predictive value of 47%. The results suggest that an oximetry study can identify children with SDB if the results are positive, but the test cannot rule out OSA. More recently, Kirk et al4 compared use of an oximetry-based home ambulatory testing device commonly used in adults to overnight polysomnography in 58 children (aged 4 to 18 years) with suspected OSA. The sensitivity and specificity were only 67% and 60%, respectively, for identifying moderate OSA (apnea-hypopnea index [AHI] >5 events/hour). Thus, oximetry testing alone is not sufficient for identifying SDB in children.
Abbreviated testing using a limited sleep study or nap studies has also been shown to be helpful in identifying SDB. Marcus et al6 showed that nap studies using sedation have a sensitivity of 74% and a positive predictive value of 100%. In a larger subsequent study, none of the individual parameters evaluated in the nap studies were predictive of abnormal overnight polysomnograms in children with SDB.7 Overall, nap studies can be helpful in identifying children with SDB but often underestimate the severity of the problem. If the abbreviated test is not diagnostic, then the child or adolescent should still have further testing to definitively evaluate for SDB.
OVERNIGHT POLYSOMNOGRAM
The ideal setting for a pediatric overnight polysomnogram would be in an environment similar to the patient’s home where the child or adolescent would feel comfortable and could sleep without difficulty. Similar to adults, preparation is essential to ensuring a successful overnight polysomnographic study. Environmental factors such as temperature, noise, bed size, and lighting control should be addressed. The child and adolescent should be adequately informed about the procedural details prior to the actual study date. Without such preparation, it is not uncommon to see either the child or parents overwhelmed by all the monitoring equipment. The patient should be at baseline status when the study is performed because even minor nasal congestion may result in overestimation of the severity of SDB. Sedation should not be used for inducing sleep in children because it can worsen airway obstruction and decrease the drive to breath, affecting the interpretation of the findings.
The ATS has published consensus statements for optimal methods for collecting data.1,2 Biocalibrations should be done when feasible. The leads, electrodes, and sensors should be sized and placed in an age-appropriate fashion. For example, leads should be passed under the clothing and secured to minimize displacement, and leads involving the head and face should be secured last.
The ideal montage for polysomnography data collection should include all of the following:
- electroencephalography channels, preferably two or more leads, C3 or C4 and O1 or O2.
- electrooculography—leads placed at 1.0 cm above and below the canthi
- chin electromyogram, leg electromyogram
- snore microphone
- respiratory airflow measurements— oral thermistor/thermocouples and nasal pressure transducer
- chest and abdominal bands for respiratory effort or motion— respiratory inductance plethysmography
- electrocardiogram
- pulse oximetry and pulse waveform (to differentiate true oxygen desaturation from motion artifact)
- end-tidal carbon dioxide monitoring (ETCO2)
- sleep position notation
Although esophageal pressure monitoring has been advocated for partial airway obstruction or upper airway resistance syndrome,8,9 these devices are not used by the majority of pediatric sleep laboratories in clinical settings.
|
|
| Table 1. Definitions of respiratory events. |
MEASURING UPPER AIRWAY OBSTRUCTION AND GAS EXCHANGE
The method of measuring upper airway obstruction is not standardized across laboratories, especially in pediatrics, and may provide different results depending on the system used. Commonly used definitions are listed in Table 1. A pneumotachograph is the gold standard for obtaining airflow data; however, it is cumbersome and requires a tight-fitting mask, which is unlikely to be tolerated by children. Most pediatric sleep laboratories prefer to use indirect airflow measurement, which is usually performed in two ways: by thermistors using temperature changes between inspired and exhaled air or by exhaled ETCO2 monitoring via side-stream capnography. The qualitative signal of thermistors provides limited information and can give inaccurate results if only nasal airflow is evaluated. Neither thermistors nor ETCO2 monitoring is ideal for measuring hypopneas or partial upper airway obstruction because the signals are qualitative and subject to interpretation. More recently, nasal pressure airflow monitoring has been evaluated in infants and children and found to be a useful adjunct in the evaluation of upper airway obstruction.10,11 Nasal pressure monitoring is more linearly related to airflow, but is affected by mouth-breathing and secretions. In such circumstances, nasal pressure monitoring may overestimate the severity of SDB.
ETCO2 monitoring is critical for determining adequate gas exchange and should be measured during pediatric polysomnography.2 Obstructive hypopneas or partial airway obstruction may occur in the absence of oxygen desaturation12-14 or arousal from sleep, but can result in elevated ETCO2. Furthermore, hypoventilation can go undetected without ETCO2 monitoring. The sampling delay in signal recording from capnography can affect the interpretation of the signal and should be assessed. Newer devices are available for measuring both ETCO2 and nasal pressure simultaneously. Oxygen saturation measurements should include pulse waveform recording to differentiate true oxygen desaturation from motion artifact. In addition, the rapid averaging time is preferred during overnight polysomnography to capture discrete reductions in the oxygen saturation that may be missed using longer averaging times. Transcutaneous monitoring of oxygen and carbon dioxide can be used as adjuncts to measuring gas exchange. A good waveform is needed to have reliable carbon dioxide data. The monitoring can be helpful for identifying trends, but has slower response times relative to the respiratory event, even with optimal skin perfusion.
NORMAL POLYSOMNOGRAPHIC VALUES
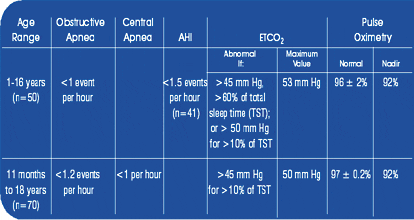
Only two studies have prospectively evaluated normal respiratory reference values during overnight polysomnography in healthy children and adolescents. The data were interpreted according to published guidelines.1 Only healthy children without symptoms of OSA were included. Exclusion criteria were obesity, airway abnormalities, any airway surgery, craniofacial anomalies, or associated lung disease. Despite some differences between the two studies, there are many similarities in the findings (Table 2).
Table 2. Normative data for respiratory events.14-16,22
Central apneas can occur during sleep, more commonly in rapid eye movement sleep. Transitional central apneas are also common. Children can have central apneas after sighs or movement arousals lasting as long as 15-20 seconds, without associated oxygen desaturation. Based on the findings, only central apneas associated with desaturation, arousal, or bradycardia are considered significant, irrespective of the duration. Most laboratories score central apneas >20 seconds duration, unless following motion or sigh.
In contrast to central apneas, obstructive apneas are uncommon in children. The pathophysiology of SDB in children includes a spectrum of airway obstruction, ranging from episodic partial airway obstruction to complete airway obstruction. In contrast, adults have less partial airway obstruction. Furthermore, in children, the partial airway obstructions may not result in arousals despite obvious changes in ventilation. Obstructive apneas are often scored differently in adults compared to children. The adult scoring criterion is >10 seconds per event; however, the studies by both Marcus et al15 and Uliel et al16 showed that apneas were short (<10 seconds and <13 seconds, respectively). Similar data were obtained in older adolescents.15 None of the studies cited duration of apnea based on age. Many centers use two respiratory cycles to calculate duration of the apnea because younger children have faster respiratory rates and adult-based criteria, may be inappropriate.1,2,10,17 Both studies15,16 found that an AHI greater than 1.2 events per hour is more than two standard deviations above normal. In contrast, adult reference values list >5 obstructive apneas per hour of sleep as abnormal.18,19 The controversy exists for adolescents because there are no specific normative data for this age group to know when pediatric or adult norms should be used to make the diagnosis of SDB.
Hypopneas are respiratory events with a reduction in airflow, but not all laboratories score these respiratory events using the same criteria in children. The American Academy of Sleep Medicine (AASM) has published consensus definitions for scoring hypopneas in adults.18,19 The two definitions that are suggested include an abnormal respiratory event lasting >10 seconds with a 50% decline in baseline airflow amplitude, or an abnormal respiratory event lasting >10 seconds with a smaller reduction in airflow amplitude, but with an associated arousal or desaturation. There are virtually no normative data in children for obstructive hypopneas. The initial study by Marcus et al15 did not include hypopneas because a standard definition for hypopnea did not exist at the time of the publication. The original data by Marcus et al15 were reviewed20 recently (N=41 of the initial 50 children) for obstructive hypopnea. Obstructive hypopnea was defined as a decrease in airflow to less than 50% baseline amplitude for a minimum of two respiratory cycles. Desaturations (>3%) and arousals were scored if present, but were not required to identify the hypopnea. Six children had any hypopneas, mean duration 12.8 seconds (range 3.5-40). One child had associated arousals with the hypopneas and two children had desaturations of 3%. The mean obstructive hypopnea index was 0.1 ± 0.1 (range 0.0-0.7) events per hour. Using the AASM definitions did not change the results. The statistically significant AHI in healthy children is therefore 1.5 events per hour (mean ±2 SD). The results show that obstructive hypopneas are uncommon in healthy children, similar to results in older adolescents.121 The limitation is that these data are based on outdated technology, and normative data using current technology (nasal pressure monitoring) are needed; however, the data contribute to the spectrum of what is considered normal.
One of the discrepancies between the two normative data studies15,16 was the ETCO2 capnography cut points. Both studies used only parameters with good waveforms. A possible explanation for the differences may be related to the type of capnometer used to collect data. The differences reiterate the paucity of data in pediatric polysomnography.
In conclusion, there are data published related to pediatric polysomnography and these provide an important reference guide for scoring respiratory parameters in children and adolescents. Although there are no population-based studies to help define clinically significant cut points in children, these data are still useful for identifying abnormalities during pediatric polysomnography. Normative data using newer technology are desperately needed to establish accurate diagnostic and clinically significant endpoints to aid in diagnosis and treatment, hence to prevent morbidity and mortality associated with SDB in children.
Manisha B. Witmans, MD, is a pediatric pulmonologist and clinical assistant professor of pediatrics at the University of Alberta, Stollery Childrens Hospital in Edmonton, Alberta, Canada; Carole L. Marcus, MBBCh, is Pediatric Pulmonologist in Sleep Medicine, Childrens Hospital of Philadelphia; Sally L. Ward, MD, is division head; and Thomas Keens, MD, is professor of pediatrics, both in the Division of Pediatric Pulmonology, Children’s Hospital Los Angeles.
REFERENCES
1. American Thoracic Society. Standards and indications for cardiopulmonary sleep studies in children. Am J Resp Crit Care Med. 1996;53:866-878.
2. American Thoracic Society. Cardiorespiratory sleep studies in children. Establishment of normative data and polysomnographic predictors of morbidity. Am J Respir Crit Care Med. 1999;160:1381-1387.
3. Brouillette RT, Morielli A, Leimanis A, Waters KA, Luciano R, Ducharme FM. Nocturnal pulse oximetry as an abbreviated testing modality for pediatric obstructive sleep apnea. Pediatrics. 2000;105:405-412.
4. Kirk VG, Bohn SG, Flemons WW, Remmers JE. Comparison of home oximetry monitoring with laboratory polysomnography in children. Chest. 2003;124:1702-1708.
5. Jacob SV, Morielli A, Morgrass MA, et al. Home testing for pediatric obstructive sleep apnea syndrome secondary to adenotonsillar hypertrophy. Pediatr Pulmonol. 1995;20:241-252.
6. Marcus CL, Keens TG, Ward SLD. Comparison of nap and overnight polysomnography in children. Pediatr Pulmonol. 1992;13:16-21.
7. Saeed MM, Keens TG, Stabile MW, Bolokowicz J, Davidson Ward SL. Should children with suspected obstructive sleep apnea and normal nap studies have overnight sleep studies? Chest. 2000;118:360-365.
8. Guilleminault C, Winkle R, Korobkin R, Simmons B. Children and nocturnal snoring: evaluation of the effects of sleep related respiratory resistive load and daytime functioning. Eur J Pediatr. 1982;139:165-171.
9. Guilleminault C, Poyares D. Arousal and UAR. Sleep Medicine. 2002;3:S15-S20.
10. Trang H, Leske V, Gaultier C. Use of nasal cannula for detecting sleep apneas and hypopneas in infants and children. Am J Respir Crit Care Med. 2002;166:464-468.
11. Witmans MB, Bohn S, Kirk VG. Reliability of nasal pressure recording during pediatric polysomnography. Am J Crit Care Med. 2003;167:A404
12. American Academy of Pediatrics. Clinical practice guidelines: diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics. 2002;109:704-712.
13. American Academy of Pediatrics. Technical report: diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics. 2002;109:1-20.
14. Witmans MB, Marcus CL, Keens TG, Ward SL. Normal polysomnographic values for obstructive hypopneas in infants and children. Sleep. 2003;26:A142.
15. Marcus CL, Omlin KJ, Basinki DJ, et al. Normal polysomnographic values for children and adolescents. Am Rev Respir Dis. 1992;146:1235-1239.
16. Uliel S, Tauman R, Greenfield M, Sivan Y. Normal polysomnographic respiratory values in children and adolescents. Chest. 2004;125:872-878.
17. Rosen CL, D’Andrea L, Haddad GG. Adult criteria for obstructive sleep apnea do not identify children with serious obstruction. Am Rev Respir Dis. 1992;146:1231-1234.
18. Meoli AL, Casey KR, Clark RW, et al. Clinical Practice Review Committee. Hypopnea in sleep-disordered breathing in adults. Sleep. 2001;24:469-470.
19. American Academy of Sleep Medicine Task Force. Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. Sleep 1999;22:667-689.
20. Witmans MB, Keens TG, Ward SL, Marcus CL. Obstructive hypopneas in children and adolescents: normal values. Am J Respir Crit Care Med. 2003;168:1540.
21. Acebo C, Millman RP, Rosenberg C, et al. Sleep, breathing, and cephalometrics in older children and young adults. Part I: normative values. Chest. 1996;109:664-672.
22. Uliel S, Tauman R, Greenfield M, Sivan Y. Normal polysomnographic values for children and adolescents. Am J Respir Crit Care Med. 2002;165:262A.