SomnoMed Ltd introduces Rest Assure, its first ever technology-enabled oral appliance.
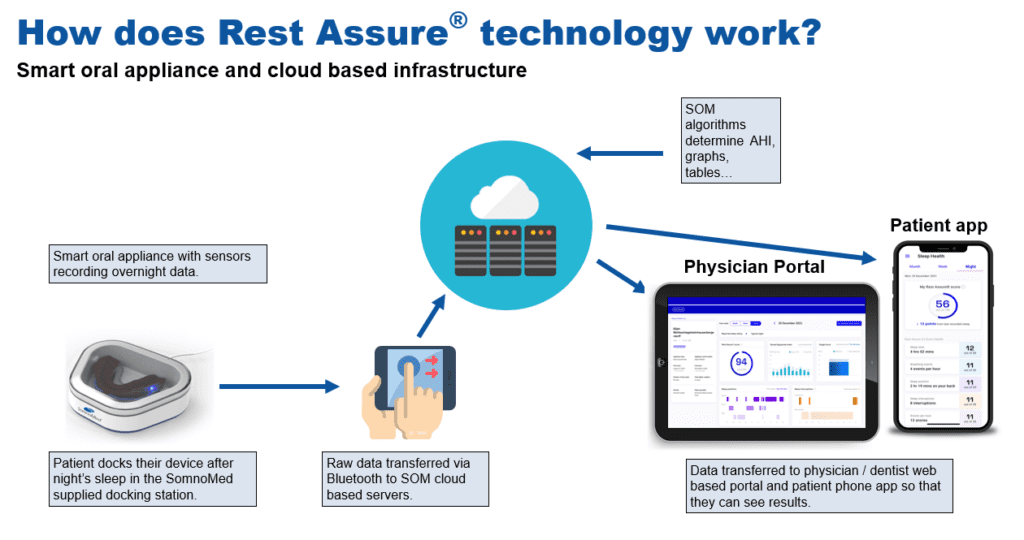
Rest Assure is a smart oral appliance that transfers efficacy and adherence data to cloud-based servers, allowing physicians and dentists to see the results. It also allows for data viewing via a patient-facing smartphone app.
Highlights of Rest Assure include:
- Addresses the lack of overnight monitoring for oral appliances.
- Sensors that measure treatment efficacy and adherence (usage) will be added to SomnoMed’s milled oral appliances.
- Physicians and dentists will be able to view personalized data from their patients on a web-based portal, alongside a patient phone app that will allow patients to view their own sleep data.
In a release, SomnoMed states the data will help support the effectiveness equation (defined by SomnoMed as: effectiveness = efficacy x compliance) and drive acknowledgement of equivalent clinical effectiveness for continuous open airway therapy (COAT) and CPAP when considering both efficacy and compliance together. Rest Assure is expected to drive prescriptions, increased reimbursement, and ultimately greater share of treatment prescriptions for COAT versus CPAP, according to SomnoMed.
In 2019, SomnoMed commissioned market research with sleep physicians that demonstrated that the lack of objective usage and efficacy data from COAT was a key barrier to prescribing oral appliances. In response, SomnoMed engaged Planet Innovation, a company specializing in medical device and software development, to assist in developing sensors, algorithms, and software to provide this data.
Using direct to patient marketing, SomnoMed expects to build brand awareness with patients who will want to interact with the Rest Assure patient app and request Rest Assure by name from their sleep physician and dentist.
The Rest Assure hardware and software is now in prototype stage, with a patient study underway to validate algorithms for efficacy and compliance. These algorithms were developed by correlating Rest Assure sensor signals and the NOX level 2 diagnostic home sleep test. The results from this validation study will be presented at the World Sleep Meeting in Rome from March 11-16, 2022 and will be included in CE mark and FDA regulatory submissions.
SomnoMed will continue to progress Rest Assure towards commercial readiness. Key milestones for the remainder of fiscal year 2022 include the completion of the patient study, final design for the docking station and sensor package, and the selection of suppliers.
In fiscal year 2023, SomnoMed will focus on the preparation and submission of regulatory documentation to the FDA, CE, and TGA. Ultimate commercialisation of Rest Assure will be subject to the review and approval by these relevant regulatory authorities.
“SomnoMed is extremely proud to bring to the sleep industry true technology that matters to patients and all treatment stakeholders,” says SomnoMed’s managing director, Neil Verdal- Austin, in a release. “We believe SomnoMed’s Rest Assure technology will empower patients to manage their own OSA [obstructive sleep apnea] treatment with a technology-enabled oral appliance providing measures of both efficacy and compliance. Whilst such data availability is standard for CPAP, having personalized treatment data, shared confidentially between clinicians and patients, is truly a first for oral appliances. We anticipate that over time, the data collected by Rest Assure will demonstrate and establish the equivalent clinical effectiveness of COAT therapy and CPAP, driving prescriptions, increased reimbursement and ultimately greater therapy market share for COAT amongst the large population of patients suffering from sleep apnea.”
Illustration 84219756 © Pratyaksa | Dreamstime.com