 |
After reflexively trying to reach and brace a car engine that swung free with too much momentum, R.T., a 38-year-old mechanic, felt a snapping sensation in his lower back and fell to the floor, unable to move. The work-related accident left R.T. with two ruptured lumbar discs (L4, L5), nerve root injury, and intermittent episodes of severe lower back pain.
When his pain was most severe, R.T. woke up nearly every time he changed positions during sleep. His pain and the resulting insomnia came under some control with medication, but soon afterward, the insomnia resumed, leaving pain clinic physicians in a quandry. Why would an otherwise healthy individual suddenly develop intractable insomnia after the successful management of a chronic pain condition? In the case of R.T., he had no previous personal or family history of insomnia. Sleep disruption due to chronic pain had been an issue, but sleep had initially improved after assessment in a pain clinic and the initiation of methadone treatment. What changed? The case of R.T. highlights the clinical value in evaluating for sleep-related central apneas compounded by narcotic medications.
BACKGROUND
Opioid/narcotic analgesic medications have been the mainstay of chronic pain therapy for hundreds of years. The term opioid refers to those medications chemically related to opium. The term narcotic refers to a broader variety of substances, often denoting a high potential for dependency. Since morphine was the first pure alkaloid to be isolated from opium in 1806, numerous morphine-like synthetic compounds have been produced. They all bind at the m-receptor subtype of the endogenous opioid receptors.
| Check the Sleep Review’s archives for more case reports. |
Insomnia symptoms associated with opioid use were first noted as early as 1981.1 In more recent years, increased risk for sleep-related central apnea has been demonstrated in patients taking narcotics, including stabilized methadone program patients.2,3 This effect is compounded by midazolam and possibly other sedative hypnotics.4 The pathogenesis of these apneas is nonspecific, and may include disturbances in regulatory activity related to medullary centers and brain stem structures, afferent influx to CNS, sleep stages, upper airways, lungs, and respiratory muscles. It is now becoming clear that these narcotic induced central apneas can lead to sleep onset insomnia and significant sleep disruption. The patient is usually unaware of any breathing problems during sleep. Also, it is difficult for the bed partner to add a corollary history, as snoring patterns may not have changed.
PATIENT CASE
After his accident, R.T. was off work for almost 6 months. Since then, he has suffered from periodic episodes of severe lower back pain. He was told he was not a surgical candidate because his injury was not causing functional limitations. He had several series of epidural steroid injections that provided only moderate relief. He was taking 30 mg of extended-release morphine (MS Contin) every 12 hours, but he continued to have severe breakthrough pain. By 2006, he was experiencing pain most of the day. He also had shooting pain down his left leg. At that point, he was again off work. He was admitted into a chronic pain program for further assessment and management.
R.T. had some difficulties sleeping, but only when his pain was most severe. He would find a comfortable position and fall asleep fairly easily. He would then awaken almost every time he adjusted his position, usually due to shooting pain. He could return to sleep rapidly, but would soon be reawakened by pain.
Several regimens of various pain medications were attempted. The doses had to be escalated, and the possibility of toxicity was considered. He was switched to methadone starting at 10 mg bid. This was gradually titrated to as high as 30 mg tid (total = 90 mg/day). Other unspecified medications were also available for breakthrough pain.
With time, his pain appeared to be well managed. However, he began to experience increasing difficulty both initiating and maintaining sleep, despite the absence of breakthrough pain. He had no notion of why he was awakening during the night. His sleep difficulties escalated to the point that the doctor at the pain clinic initiated a referral to the sleep clinic.
DIAGNOSING THE PROBLEM
R.T. has been steadily employed for more than 12 years and has been married for 14 years. He has three children, aged 4, 6, and 10 years. Other than typical financial concerns, there are no sources of stress in the family environment.
Up until his work accident in September 2004, R.T. had been in perfect health. He engaged in regular physical exercise at a local fitness club. He was also quite active in his work.
Since his back injury, he has been unable to work consistently. He has been on a variety of medications, with limited success. He has also been attending a physiotherapy clinic on a weekly basis, but this has also been a limited success.
Since commencing with methadone treatment, his pain has come under some control, and he is able to work more regularly. However, his daytime functioning is now impaired by intractable sleep disruption, and he finds it difficult to concentrate on most days. Due to his physical limitations, he is now responsible for more sedentary duties at work. He has actually fallen asleep at his desk, and his boss has challenged him on more than one occasion.
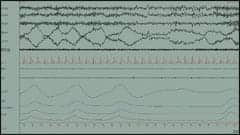 |
| Figure 1. Sleep-onset central apnea. |
R.T. drinks about two to three caffeinated beverages per day. He has eliminated alcohol consumption since commencing with opioid therapy. He is a nonsmoker. His exercise is now limited to modest work activity and physiotherapy routines. His weight has increased by approximately 40 pounds since his injury. His medical history is unremarkable, other than issues related to his back injury. Recent complete blood work and 12-lead ECG were entirely normal.
R.T. was seen in consultation in the sleep clinic. He completed a comprehensive Sleep-Wake Questionnaire prior to this appointment, as well as two psychological self-rating questionnaires—the Beck Depression Inventory (BDI) and the Symptoms Checklist (SCL-90).
SLEEP HISTORY
R.T. gets into bed between 10:30 and 11:30 pm. Sleep onset is often delayed for 30 to 120 minutes. He does not engage in circular thinking or anxious rumination prior to sleep onset. He has no notion of what keeps him from getting to sleep. Once sleep does occur, he awakens frequently throughout the night. Awakenings are almost always spontaneous. His wife does not complain of significant snoring, and she has not noted significant restlessness before or during sleep. She has noticed respiratory pauses, with some episodes of resuscitative breathing.
He usually arises at 6 am while working (7:30 am on days off). He estimated his total sleep time to be 5 to 6 hours per night, on average. He denies morning headaches. He denies experiencing a dry mouth or a sore throat upon awakening. He has some difficulty arising in the morning, but is “OK” once he gets going. He reports only mild daytime sleepiness, but does admit to occasional inadvertent naps. Even at his best, he never feels rested, and he struggles every day to perform adequately at work.
PSYCHOLOGICAL SELF-RATING QUESTIONNAIRES
R.T. scored 17 on his Beck Depression Inventory, which indicates mild emotional distress. However, most of his symptoms were more somatic than emotional. On his Symptoms Checklist, he endorsed moderate anxiety, but denied symptoms of significant depression. During his clinical assessment, he reported that his anxiety was related specifically to his inability to perform his various family and work duties due to his intractable back pain. He was also quite certain that his insomnia was exaggerating his emotional symptoms.
SLEEP STUDY
Due to his wife’s observation of pauses in respiration, as well as his unexplained sleep disruption, a sleep study was indicated.
Sleep EEG: The study commenced at 22:50 hours. Sleep onset was delayed for 115 minutes. His sleep was fairly well consolidated, until a period of wakefulness between 03:15 and 04:20 hours. Sleep architecture was marked by frequent EEG arousals and the complete absence of slow wave sleep. His sleep efficiency index was markedly reduced at 66%.
Respiration: Respiratory recordings showed sleep-onset central apneas and hypopneas, and these appeared to contribute to a delayed onset to sleep. During sleep, there were 309 central apneas, 48 obstructive apneas, and 59 hypopneas noted, for a respiratory disturbance index of 77 events/hour, associated with occasional mild snoring, frequent respiratory arousals, and moderate arterial oxygen desaturation to as low as 77%.
Movements: There was no evidence for significant motor restlessness while lying awake in bed or periodic leg movements during sleep.
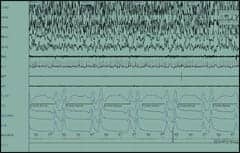 |
| Figure 2. Narcotic-induced central apneas during sleep. |
TREATMENT
Upon reviewing the results of the sleep study, we suspected that the central apneas were being exacerbated by his narcotic analgesic medications. We contacted the referring physician at the pain clinic, and it was agreed that gradual downward titration of the methadone was a reasonable strategy. The goal was to reduce the medication to an ideal dose where pain relief was sustained, in the absence of significant nocturnal respiratory depression.
R.T.’s pain medications were titrated to 20 mg bid (total dose = 40 mg/day) over a 2-week period, with doses being taken earlier in the day. Although the frequency of breakthrough pain episodes increased, his insomnia improved significantly, without the use of sedative hypnotic medications.
With prominent central apneas, we did consider treatment with a variable positive airway pressure (VPAP). These devices have recently been approved for the treatment of central sleep apnea, mixed sleep apnea, and periodic breathing. Given the severity of his central apnea, he would be a good candidate had his central apnea not improved after tapering his methadone.
REPEAT SLEEP STUDY
Over time, R.T. once again began to show difficulty maintaining sleep. When he was seen in follow-up, he attributed this mainly to breakthrough pain during sleep, probably related to his much lower dose of methadone. An additional baseline study was conducted for comparison, especially with regard to sleep-related respiration.
Sleep EEG: The study commenced at 23:45 hours. Sleep onset occurred within 32 minutes. Moderate sleep fragmentation was displayed, but all sleep stages were normally represented, except for a reduction of slow wave sleep at 6.2% of total sleep time (normal range = 10-15%). His sleep efficiency index was normal at 87%.
Respiration: Respiratory recordings showed 37 obstructive apneas, 18 central apneas, and 56 hypopneas, for a respiratory disturbance index of 17 events/hour, associated with occasional mild snoring, occasional respiratory arousals, and occasional very mild arterial oxygen desaturation to as low as 88%.
Movements: There was no evidence for significant motor restlessness while lying awake in bed or periodic leg movements during sleep.
DISCUSSION
This patient may have a predisposition for unstable airway control, and thus may have been somewhat more sensitive to the effects of narcotic medications. It is clear that not all patients experience significant sleep problems with narcotics on board, and the threshold dose for a significant effect is likely different for every patient. The key with this patient was working cooperatively with the pain management group to titrate his methadone to lower doses until sleep improved. Unfortunately, this has to be balanced with the sleep-disruptive effect of breakthrough pain.
Methadone has unique properties that make it an attractive option in the treatment of complex pain syndromes such as neuropathic pain. Historically, it has been a significant challenge for pain clinicians to manage chronic severe pain, due to the high doses needed when using “traditional” opioids such as morphine, hydromorphone, and fentanyl. In our clinic, we have found the longer-acting opioids have a more profound effect on sleep-related breathing. MS Contin, fentanyl patches, and methadone are among the worst offenders.
We now get very appropriate referrals from this particular pain clinic, as they are now aware of the increased risk of sleep-related consequences when using narcotics for the management of chronic pain syndromes. The gravity of this message becomes especially apparent considering that these patients often receive a sedative-hypnotic medication for the management of their emerging insomnia complaints. The combination of a high dose narcotic analgesic (causing central suppression of respiratory drive) with a sedative-hypnotic medication (for the management of the resultant sleep disruption) could have deleterious consequences.
James MacFarlane, PhD, DABSM, is assistant professor, Departments of Paediatrics and Psychiatry, University of Toronto. He is also director of education at the Toronto Sleep Institute and a consultant at the Niagara Sleep and Snoring Centre. He can be reached at [email protected].
REFERENCES
- Kay DC, Pickworth WB, Neider GL. Morphine-like insomnia from heroin in nondependent human addicts. Br J Clin Pharmacol. 1981;11(2):159-169.
- Bailey PL, Pace NL, Ashburn MA, Moll JW, East KA, Stanley TH. Frequent hypoxemia and apnea after sedation with midazolam and fentanyl. Anesthesiology. 1990;73(5):826-30
- Farney RJ, Walker JM, Cloward TV, Rhondeau S. Sleep-disordered breathing associated with long-term opioid therapy. Chest. 2003;123(2):632-639.
- Wang D, Teichtahl H, Drummer O, et al. Central sleep apnea in stable methadone maintenance treatment patients. Chest. 2005;128(3):1348-56.






