Medical device sleep apnea and snoring disorder company Oventus Medical Ltd releases its first 4E. The company states it has established a platform for accelerated growth following a successful IPO raising $12 million and is reporting encouraging early revenue.
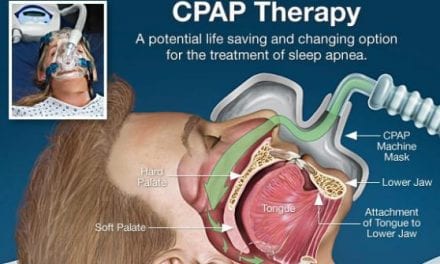
Oventus is focused on developing and producing oral appliances to treat obstructive sleep apnea and snoring, especially in people who cannot be, or are not effectively treated with existing devices or therapies.
CEO Neil Anderson says in a release, “The company’s devices are 3-D printed and feature a unique airway that channels air to the back of the throat, bypassing oral obstructions that cause sleep apnea and snoring. We have established state of the art systems and processes that will drive growth for the company going forward.”
Oventus has reported early revenues of $540,164 for the 12 months to June 30, 2016.
The company received clearance from the US Food and Drug Administration (FDA) for its first generation device, the O2Vent Mono, and has made an application for its second generation titratable device, the O2Vent T.
Oventus launched the O2Vent T in Australia and enrolled the first patient in a 40 patient clinical trial of the device. The trial will produce data around the comfort, safety and efficacy of the O2Vent T, as well as the comfort and efficacy of the inclusion of an airway into an oral appliance. Oventus has a unique position with a patent protected device in the growing sleep disorder market.
A scientific advisory committee of sleep, dentistry, and ear, nose, and throat experts has been established in Australia to advise on clinical trial design for Oventus’ current and future products.
Clinical director and founder Chris Hart, M.Phil, BSc BDSc, says; “We believe obstructive sleep apnea is a serious medical condition that requires a multidisciplinary approach to treatment. We look forward to working with the committee so that our devices achieve maximum patient benefit.”
Oventus has also established a 3-D titanium printing facility at CSIRO in Melbourne and a production operation in Brisbane for polymer inserts and packaging.