The simple facts about recognizing and managing complex sleep apnea.
One of the most rewarding aspects of sleep medicine is the experience of speeding a severely symptomatic patient with sleep apnea through sleep consultation, successful split-study polysomnogram (PSG), rapid initiation of positive airway pressure (PAP) therapy, and near-complete resolution of symptoms. After more than 20 years of evaluating and treating such patients, I still get a thrill out of such a completely successful scenario. The patient’s big smile during the first follow-up office visit is the best part. Frequently, PAP therapy can make literally an overnight difference. Try doing that with far-advanced chronic obstructive pulmonary disease (COPD) or severe pulmonary fibrosis.
In contrast, one of the lows of sleep medicine is when patients cannot be effectively stabilized and return to the office wearing a scowl instead of a smile. When the patient still has eyelids that need to be propped open with toothpicks after starting PAP therapy, we know we are in trouble. I have certainly seen more than my share of this latter group. In fact, for several years I had become more and more frustrated with my own and my colleagues’ inability to effectively treat what seemed to be a larger and larger patient group.
BACKGROUND
The patients our sleep disorders center (SDC) staff experienced the most trouble with often had mixed or central events during titration PSGs. Some of the patients had chronic heart failure (CHF) with the usual regular waxing and waning of Cheyne-Stokes respiration (CSR). However, many other patients had central events that were less regular, more chaotic, and intermingled with mixed or obstructive events. Apneas and sleep fragmentation seemed to persist regardless of what we did. Our technologists would increase and decrease the CPAP device’s pressure setting during our titrations. When CPAP failed, they would convert to bilevel spontaneous (bilevel S) therapy, and when that failed to stabilize the patients, we would bring them back for repeat titrations and try bilevel spontaneous/timed (bilevel ST) treatment, entrainment of O2, addition of hypnotic or sedating agents (to decrease respiratory drive), and/or avoidance of the supine sleep posture. Often no treatment was effective, and sometimes our extra manipulations seemed only to make our patients worse than they had been with simple CPAP alone or even with no treatment at all.
We had read about a new PAP modality—adaptive servo-ventilation (ASV)—that had been used in Europe for central sleep apnea (CSA) and CSR in patients with CHF.1 However, the device was unavailable in the United States. We wondered if that modality would help treat our difficult patients who did not have CHF with CSA/CSR.
We began to question our long-held assumptions about how to treat patients with mixed or central events during titration PSGs. During the latter part of 2004, we decided to treat only obstructive events during a first titration PSG (or the titration half of a first split-study). We would leave the central events untreated and would decide individually for each patient (after PSG interpretation) whether to attempt further treatment during a second titration PSG.
ARTICLES LEAD TO NEW INSIGHT
Several months after we stopped titrating for central events, we read a report2 documenting that bilevel therapy often did worsen central events. We were not alone in our struggle with these patients!
While the article did not offer alternative treatment strategies, it was comforting. Another publication3 explained more about the subtypes of CSA (Sidebar 1). This paper was helpful, as were other subsequent reports.4-6 We were watching the birth of a new syndrome: complex sleep apnea (CompSA). Our problematic patients were part of a larger family of patients that other SDCs also found difficult to stabilize. The mechanisms are complex and incompletely understood, but investigators are teasing out some important factors (Sidebar 2).
TRADITIONAL AND INNOVATIVE TREATMENTS FOR CSA
Treatment recommendations for patients with the different types of CSA vary because, at present, there is insufficient published data for reliable clinical guidance. Several reports have demonstrated little benefit, no change, or worsening of CompSA with CPAP or bilevel therapy.7-10 The researchers behind a large Canadian multicenter controlled randomized trial of CPAP use in CHF and CSA/CSR recently terminated their study early because of lack of efficacy.11
Some have advocated treating CSA with supplemental oxygen. However, the benefit of oxygen has been equivocal or incomplete at best.12 Pharmacologic agents also have been considered. The drugs theophylline and acetazolamide are known to improve periodic breathing, but adverse effects (arrhythmias, worsening of obstructive events) limit the utility of these drugs. Moreover, there are no data evaluating these drugs as part of a multimodality treatment regimen.13,14 While the use of drugs that promote stable non-REM sleep is attractive,3 there are little, if any, data supporting their use. Drugs that are known to induce or worsen CompSA, such as high-dose long-acting opioids,15 should logically be minimized or avoided entirely, if possible.
A partial treatment for CSA that appears to have some merit is body-position modulation.16 While this technique has been effective for CSA/CSR patients, there is little data evaluating this treatment in patients with CompSA.
Because the above traditional treatments have been largely ineffective, patients with CompSA often undergo repeated PSGs with multiple therapeutic manipulations in an attempt to stabilize them more effectively. Typically, the patients do poorly with any treatment. For this reason, they are frequently noncompliant with therapy. A common scenario is that either the patient or the physician gives up, and the patient remains at risk because of absent/ineffective treatment.
A critical component of therapy in these patients is minimizing or eliminating the CO2 homeostasis instability. The use of nonvented masks may be effective treatment in patients with mild CompSA. In more severe cases, some advocate the addition of dead space (either alone or in conjunction with PAP therapy).3 Small increases in inhaled CO2 have been achieved experimentally using a prototype device, the PAP Gas Modulator.3,4 These treatments appear promising but should presently be considered experimental.
ASV BECOMES AVAILABLE
European and Australian sleep physicians and therapists have used ASV for the past few years to successfully treat several thousand patients with CSA/CSR secondary to CHF. However, researchers have not extensively evaluated the efficacy of ASV in other types of CSA.
When we first read about the technique,1 we were fascinated by the concept and wanted to try ASV for ourselves. Our efforts to acquire a prerelease device from the manufacturer were unsuccessful, however.
When subsequent reports17,18 confirmed the findings in the first study we had read and showed that the ASV device remained effective for study periods of up to 6 months, our interest only increased. We again wondered if ASV might not be an answer for some of our “difficult” patients.
Late in 2005, the US Food and Drug Administration (FDA) finally approved the VPAP Adapt SV™ (Adapt SV) device for use in adults with respiratory insufficiency caused by the various kinds of central and/or mixed apneas and periodic breathing. The manufacturer, ResMed, contacted us and said we could have one of the 13 devices it was providing for a controlled market release to interested SDCs. We would finally get our chance to see if ASV might help our difficult patients.
Since the device had just been approved, no formal clinical indication criteria had been delineated and there was no established billing pathway. The Centers for Medicare and Medicaid Services (CMS) had yet to decide whether it would authorize use of the device. We had many questions about the effectiveness of the product, the pathway for payment, and whether patients would accept the device and find it helpful.
WHAT IS ADAPTIVE SERVO-VENTILATION?
ASV is a novel form of bilevel ventilation. The pressure provided during inspiration varies, increasing when the patient’s tidal volume decreases, and conversely. This varying assist helps to regularize the chaotic pattern of respiration by reducing and then eliminating the alternating periods of patient over- and under-breathing. This feature operates rapidly, within two or three breaths (almost functioning like a rapidly acting adaptive PAP or A-PAP device). The ASV algorithm also monitors recent average minute ventilation (using the trailing 3-minute ventilation) and continuously calculates a target ventilation (90% of the recent average ventilation). This relatively weak, but persistent, downward constraining force on minute ventilation serves to minimize/eliminate the CO2 instability and raises the PaCO2 just enough to prevent the frequent CO2 dips below the apnea threshold. A “smart” backup rate is actuated automatically only if/when needed. The expiratory PAP or EPAP-equivalent is called the “EEP” (end-expiratory pressure) as the ASV waveform is “softer,” meaning that it decreases more slowly during exhalation than traditional bilevel devices that have a near-square waveform. The default EEP is 5 cm H2O, and can be raised to a maximum of 10 cm H2O. The default pressure support (the inspiratory PAP or IPAP equivalent) is 3 cm H2O greater than the EEP, but can be adjusted upward if needed.
OUR EARLY EXPERIENCE
By the time we acquired the device, we had identified a challenging first subject. The patient had a 2-year history of sleep-disordered breathing (SDB) with an apnea hypopnea index (AHI) of 40. He had been doing well with CPAP until he sustained an intracranial aneurysm rupture. After this episode, he had difficulty using CPAP and his sleep quality deteriorated. A repeat diagnostic PSG revealed severe worsening of his OSA (more than 90% were obstructive events) with an AHI = 114/h. A subsequent CPAP PSG revealed an increase in the AHI to 123/h with a change to more than 90% central events! The patient represented a classic example of the emergence/worsening of central events with PAP, which is essentially the definition of CompSA. We had never seen such a large shift from obstructive to central events with PAP. I was almost certain the ASV device would be no match for this patient.
On the night of the study, our control room was full of interested observers. We crowded around the monitor with anticipation. The patient fell asleep rapidly, and we began the trial at the device’s default settings. I was not surprised when severe obstructive events continued during the first 1 to 2 hours of the study, but I was encouraged that the central events virtually disappeared almost immediately.
As the night progressed, we slowly and cautiously increased the EEP. I left the SDC at around 2 am when the patient was clearly much better, although still not completely stabilized. By the end of the night, the techs had increased the EEP to the maximum of 10 cm H2O, and the AHI had nearly normalized to 6/h. Only some modest hypoxemia and a few obstructive hypopneas persisted. We were very surprised, and also very pleased, to see such a spectacular first-patient response. When our second patient also was effectively stabilized (this time at the default EEP of 5 cm H2O), we knew we were experiencing more than just beginner’s luck, and believed that we had found a device that was going to make a real difference for many of our most difficult patients.
We have used ASV for longer than a year now and have evaluated more than 20 patients. Many of these patients currently use ASV regularly, and almost all report a significant positive difference compared to CPAP. In general, the patients with the most severe underlying SDB have experienced the greatest benefit. We have studied patients with large variations in age, body mass index (BMI), Epworth score, AHI, and percentage of central events during the diagnostic PSG. Common underlying conditions have included: hypertension, atrial fibrillation, coronary artery disease, mild left ventricular dysfunction (none of our patients has had overt CHF), chronic pain with regular use of long-acting opioids, diabetes mellitus, and hyperlipidemia; one young patient had myotonic dystrophy. A number of patients have had several of the above diagnoses.
We initially thought we would be evaluating ASV therapy primarily in patients with heart failure and CSA/CSR, but our cardiologists are referring just a few such patients. We have therefore become more interested in the patients referred to the SDC that fail traditional treatment, indicating that they may have CompSA. We have focused our attention on this patient population while we continue to help educate our cardiologists about the relationship between CHF and SDB.
The average AHI in the patients we have studied to date has been about 45/h. With CPAP or bilevel therapy, the patients’ AHIs decreased only modestly, but with ASV, the AHIs have normalized in about 80% to 85% of our patients. Typically, the device seems to eliminate, or nearly eliminate, events of all types. Initially, we were hesitant to increase the EEP too rapidly; with experience, we have become more aggressive with our titrations.
As the device is minute ventilation-driven, the manufacturer recommends using it with full-face masks to include any oral flow in the calculated ventilation. In several patients, ineffective mask fit, persistent leak, or patient intolerance required us to change to a nasal interface, and we found that this also was usually effective.
There is a learning curve for the technologists. For the routine patient, the titration is extremely simple. However, extra skill and experience are required for some of the more difficult patients. Based on our experience, we suggest that initially only a small core group of a SDC’s most experienced technologists become involved with ASV.
In addition, we found that sometimes it was difficult to determine during a titration whether a real event has occurred as the rapidly responding pressure support may terminate a near-event just before the 10-second mark. In one patient, we initially overtitrated the EEP, increasing the pressure for respiratory effort-related arousals (RERAs) that were, on formal scoring, demonstrated to be spontaneous arousals. In another patient, undertitration occurred as the technologist adjusted the EEP for apneas and hypopneas, but did not adequately increase the pressure for residual RERAs. Our experience suggests that some of the early failures with ASV may be inadequate SDC/technologist experience, and not necessarily a problem with the device.
Our results are similar to those found both in a single case report by Peter Gay, MD,19 and also to a series of 21 patients reported by Timothy Morgenthaler and colleagues.20 In the latter study, patients who failed to stabilize with CPAP underwent a comparative evaluation with ASV vs noninvasive positive pressure therapy using bilevel ST. Of the nine patients with CompSA in that study, the mean untreated AHI was 49/h, and decreased minimally with CPAP to a still very elevated 42/h. With NPPV, the AHI decreased substantially further to 6.8/h. Even this improvement, however, was inferior to the results with ASV when the AHI decreased further to 1.6/h. ASV was similarly effective in the other groups (CSA/CSR, predominantly mixed apneas) evaluated in the study.
SCOPE AND LIMITATIONS OF ASV
The complete scope and any limitations to successful treatment with ASV remain to be defined. Obstructive events, if severe, may persist in some patients because of insufficient airway splinting at the maximum EEP. In these patients, bilevel ST with an EPAP greater than 10 cm H2O may be the treatment of choice. One of our patients appeared to stabilize at maximum EEP only in the lateral posture. Typically, the incompletely stabilized patients have a high BMI and have CompSA that might be described as being superimposed on a base of severe OSA. The presence of patient-ventilator dyssynchrony has been reported with ASV under certain circumstances, when additional measurements of flow and pressure are recorded, and/or when sophisticated experimental ECG analysis techniques are used to evaluate differences in cardiopulmonary coupling3 (personal communication with Robert J. Thomas, MD).
As is the case with all forms of PAP, successful ASV treatment is occasionally difficult in patients with an underlying bias against PAP therapy and in patients with poor sleep hygiene or insomnia. Moreover, ASV does not treat sleep fragmentation from limb movements or spontaneous arousals. Hence, we had a few patients whose sleep remained fragmented despite effective ASV elimination of SDB events. We also had one patient in whom modest hypoxemia persisted despite ASV and required O2 entrainment.
Our wish list for the next version of the Adapt SV includes a higher maximum EEP and the ability to download apnea and hypopnea data from the device. We have come to rely heavily on the autoscoring data that now can be accessed on modern CPAP and bilevel devices in our daily management and trouble-shooting of our patients with SDB. Hence, it is unfortunate that the FDA precluded the use of this feature at the time of the initial approval of the device. We hope that this feature will soon be restored.
We had two or three patients for whom we could not determine with certainty the optimal EEP during a single-night PSG. The ability to record event-frequency data over several/many nights would be very helpful in optimizing therapy in these patients. The external pressure line, which measures pressure at the mask-end of the hose and is attached to the outside of the main hose with a series of clips, is somewhat cumbersome. If this line could eventually be eliminated or incorporated into the hose (this was done years ago in a Healthdyne CPAP device), the device would have a sleeker look and would be a bit easier to use. By today’s standards, the device also is a bit large and heavy, although its complexity may well preclude the possibility of allowing it to fit into a smaller case. Finally, we initially found downloading data from the device difficult, and although recent software upgrades have simplified this process, downloading data remains a bit time-consuming and requires a cable connection to the device; no card-reader download is presently available.
SUMMARY
We remain very enthusiastic about ASV as our experience and our patient base expand. In our opinion, development of the Adapt SV represents a major step forward in the treatment of CompSA. This may be the most significant improvement in PAP therapy since the original development of bilevel therapy. We expect that numerous clinical trials (some undoubtedly already in progress) will further demonstrate the scope and limitations of this intriguing technique over the next several years. The role of ASV in CompSA may equal or in fact eclipse the usefulness of the device in CSA/CSR associated with CHF, which was the initial target market for the device.
Competition between device manufacturers also may encourage further improvements in the technology. On March 7, Respironics Inc, Murrysville, Pa, announced that the FDA granted 510(k) clearance to its BiPAP autoSV device for treating central and/or mixed apneas and periodic breathing. Other manufacturers will undoubtedly develop similar devices, too, in an attempt to capitalize on this market, which some investigators believe may be as large as 10% to 15% of the existing sleep apnea population. From our limited experience to date, we think these estimates may be high, although perhaps our current criteria for diagnosis are too stringent.
We have been pleased with our experience with the Adapt SV device and look forward to collecting experience with the BiPAP autoSV device and any other ASV devices that may enter the market. If your experience mirrors ours, you too will soon be able to say that this new technology makes many “complex” patients “simple.”
Stephen E. Brown, MD, DABSM, is medical director of Memorial Care Sleep Disorders Centers in Long Beach, Calif. Also contributing technical assistance with the article were Kerry Kouchi, RPSGT; Tamera Godfrey, RPSGT; Heather Kronstedt, RPSGT; and Daniel Brennan, RPSGT. He and his colleagues may be contacted by e-mail at [email protected].
CASE REPORT
A 48-year-old disabled man with poor sleep hygiene, a “free-running” sleep-wake cycle, hypertension, and chronic use of long-acting opioids due to severe back pain with multiple surgeries presented to the sleep center with snoring, witnessed apneas, nonrestorative sleep, and insomnia suggesting sleep-disordered breathing (SDB). The patient also had poor sleep hygiene, given his nearly free-running wake-sleep schedule. A diagnostic polysomnogram (PSG) confirmed that he had an apnea hypopnea index (AHI) of 104/h with 70% central events. Sleep fragmentation was severe, and the sleep efficiency was greatly reduced (Figure 1).
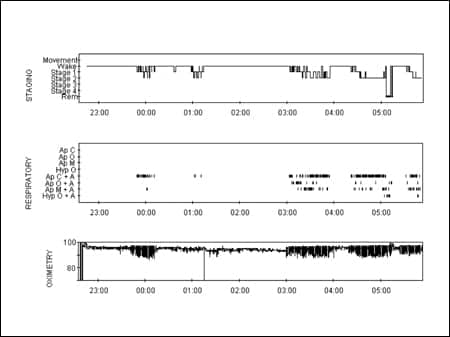 |
| Figure 1. The diagnostic polysomnogram. |
The patient responded poorly to CPAP; sleep fragmentation continued to be severe, and the AHI remained moderately/severely increased (see Figure 2). We diagnosed complex sleep apnea (CompSA); although some investigators consider central apnea caused by chronic opioid therapy a different disorder, we like to consider it a subtype of CompSA. He underwent an ASV titration and was better stabilized by the end of the night (Figure 3). However, his sleep efficiency remined low until we established the effective end-expiratory pressure (EEP) toward the end of the night. When finally stabilized with an AHI = 0/h, his sleep efficiency normalized and a REM rebound occurred.
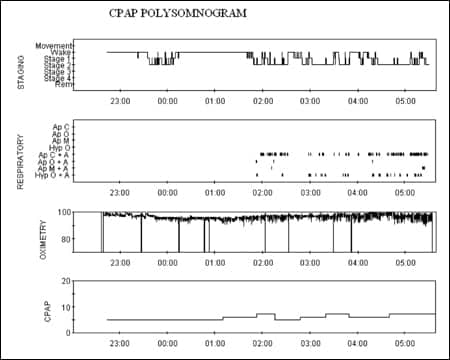 |
| Figure 2. Polysomnogram data showing the patient’s poor response to CPAP. |
The patient’s initial subjective response to ASV was good. After more than 9 months of quite regular ASV use (both self-reported and from the compliance monitoring built into the device), he said he was now often swimming 3,000 yards, something he had been unable to do previously, and was sufficiently pleased with his outcome to give written consent to have his case presented.
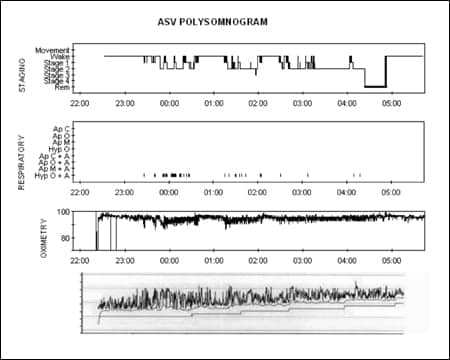 |
| Figure 3. Polysomnogram data showing the patient’s improved response to ASV. |
We selected this patient as an average ASV responder. His case illustrates that patients with CompSA may also have related problems that can complicate therapy (poor sleep hygiene and insomnia). In addition, this case shows that many patients with CompSA will not necessarily be stabilized at the default EEP of 5 cm H2O and will need titration to higher pressures.
References
- Teschler H, Dohring, J, Wang YM, Berthon-Jones M. Adaptive pressure support servo-ventilation: a novel treatment for Cheyne-Stokes respiration in heart failure. Am J Respir Crit Care Med. 2001;164(4):614-619.
- Johnson KG, Johnson DC. Bilevel positive airway pressure worsens central apneas during sleep. Chest. 2005;128(4):2141-2150.
- Gilmartin GS, Daly RW, Thomas RJ. Recognition and management of complex sleep-disordered breathing. Curr Opin Pulm Med. 2005;11(6):485-493.
- Thomas RJ, Daly RW, Weiss JW. Low-concentration carbon dioxide is an effective adjunct to positive airway pressure in the treatment of refractory mixed central and obstructive sleep-disordered breathing. Sleep. 2005;28(1):69-77.
- Pusalavidyasagar SS, Olson EJ, Gay PC, Morgenthaler TI. Treatment of complex sleep apnea syndrome: a retrospective comparative review. Sleep Med. 2006;7(6):474-479.
- Morgenthaler TI, Kagramanov V, Hanak V, Decker PA. Complex sleep apnea syndrome: is it a unique clinical syndrome? Sleep. 2006;29(9):1203-1209.
- Sin DD, Logan AG, Fitzgerald FS, Liu PP, Bradley TD. Effects of continuous positive airway pressure on cardiovascular outcomes in heart failure patients with and without Cheyne-Stokes respiration. Circulation. 2000;102(1):61-66.
- Guilleminault C, Stoohs R, Labanowski M, Simmons J, Clerk A. Cardiac failure, snoring ventricular arrhythmias and nasal bilevel positive pressure ventilation. Sleep. 1993:16(8 suppl):S139-S140.
- Buckle P, Millar T, Kryger M. The effect of short-term nasal CPAP on Cheyne-Stokes respiration in congestive heart failure. Chest. 1992;102(1):31-35.
- Davies JR, Harrington KJ, Ormerod OJ, Stradlin JR. Nasal continuous positive airway pressure in chronic heart failure with sleep-disordered breathing. Am Rev Respir Dis. 1993;147(3):630-634.
- Bradley TD, Logan AG, Kimoff RJ, et al. Continuous positive airway pressure for central sleep apnea and heart failure. N Engl J Med. 2005;353(19):2025-2033.
- Thalhofer SA, Dorow P, Meissner P. Influence of low-flow oxygen supply on sleep architecture in patients with severe heart failure (NYHA III-IV) and Cheyne-Stokes respiration. Sleep Breath. 2000;4:113-120.
- Orth MM, Grootoonk S, Duchna HW, et al. Short-term effects of oral theophylline in addition to CPAP in mild to moderate OSAS. Respir Med. 2005;99:471-476.
- Hu K, Li Q, Yang J, et al. The effect of theophylline on sleep-disordered breathing in patients with stable chronic congestive heart failure. Chin Med J (Engl). 2003;116:1711-1716.
- Farney RJ, Walker JM, Cloward TV, Rhondeau S. Sleep-disordered breathing associated with long-term opioid therapy. Chest. 2003;123:632-639.
- Szollosi I, Roebuck T, Thompson B, Naughton MT. Lateral sleeping position reduces severity of central sleep apnea/Cheyne-Stokes respiration. Sleep. 2006;29(8):1045-1051.
- Pepperell JCT, Maskell NA, Jones, DR, et al. A randomized controlled trial of adaptive ventilation for Cheyne-Stokes breathing in heart failure. Am J Resp Crit Care Med. 2003;168:1109-1114.
- Philippe C, Stoica-Herman M, Drouot X, et al. Compliance with and effectiveness of adaptive servo-ventilation versus continuous positive airway pressure in the treatment of Cheyne-Stokes respiration in heart failure over a six month period. Heart. 2006;92(3):337-342.
- Gay, PC. More than just OSA: the clinical challenge of treating complex sleep apnea. Sleep Review. 2006;7(6):30-33.
- Morgenthaler TI, Gay PC, Gordon N, Brown LK. Adaptive servoventilation versus noninvasive positive pressure ventilation for central, mixed, and complex sleep apnea syndromes. Sleep. 2007;30(4):468-475.
- Eckert DJ, Jordan AS, Pankaj M, Malhotra A. Central sleep apnea—patho-physiology and treatment. Chest. 2007;131:595-607.