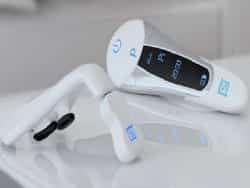
SnooZeal an over-the-counter smart phone controlled anti-snoring/sleep apnea medical device that its manufacturer states is aimed at reversing the “root cause of snoring and obstructive sleep apnea.” The app, used in conjunction with the device, allows users to monitor improvement daily. SnooZeal’s technology sends gentle electronic pulses to the back of the tongue to tone the muscles, which the manufacturer states reverses the over-relaxation of the tongue and stops snoring.
The SnooZeal device is used during the day only and is worn for 20-25 minutes, twice a day over a 6-week period.
Developed by ear-nose-throat consultant Anshul Sama, BMedSci, BM, BS, FRCS, FRCS, senior consultant surgeon at Nottingham University, SnooZeal addresses the root cause of excessive relaxation and loss of tongue muscle tone during sleep. Effectively giving the tongue a “work-out” it uses wireless technology to exercise the tongue to treat the physiological cause of snoring and sleep apnea. The smart phone app monitors the user and controls the progress of the treatment.
“In the US alone, over 90 million Americans snore every night, not only disturbing their sleep but that of their partners too. Noisy snoring strains relationships as it disrupts the partners sleep more than the snorers. It is well known that the long-term effects of sleep deprivation and disturbed sleep can lead to serious health problems,” Sama says in a release.
Since receiving CE approval earlier this month, the team behind SnooZeal are working on European pre-sales with retailers, ENT consultants, medical device distributors, and sleep physicians. SnooZeal has a patent granted and has retained ISO13485. The company states that FDA approval is expected soon, European trial results are equally awaited, and University College Hospital London is commencing trials in January and will be available over the counter in 2017.
“Many nighttime anti-snoring/sleep apnea devices are often abandoned as people find them uncomfortable and ineffective. What makes SnooZeal so innovative and effective is that it addresses the root cause of snoring rather than just temporally relieving it, and because SnooZeal is only used during the day, it means that users can sleep naturally at night,” says Sama.



This is a true invention, I suffer from OSA and Snoring both… and the mask feels like I’m in ICU.. about to die!!
Especially the pressure from my mask is exerted to much that I feel claustrophobic!
This will be a true life saver… wonder where I can get this?
Hello Steve,
Thanks for your comment. Snoozeal just finished our first clinical study which showed a reduction in snoring, mild, moderate and even OSA by 67%. We are in the process of recruiting patients for a large study (over 1,000 patients) with the device. Please let me know if you are interested and I will add you to the list.
Sincerely,
Wayne
Investor Snoozeal Inc.
Can I purchase this in the USA ?
If not when will it be available?
As the report indicates US FDA Clearance is pending, I would be very interested in discussing marketing the Snoozeal in the US. We are the premier comprehensive importer/distributor providing solutions to Sleep focused ENT practices in the US. Our products/services range from diagnosis to therapy to surgery, and this would complement our portfolio very well.
I also have a problem with snoring and wonder if I would qualify to trial your device?
I also suffer from snoring and wonder if I would be able to qualify in your trials.
Dear Wayne,
As a narcolepsy patient organization we have several people in our test panel who would love to join your trials. Please let me know if I can interest you in having these people apply and how they should do that. The narcolepsy community has many people with snoring as a secondary problem and a positive outcome from our panel will surely help future sales.
Kind regards,
Feri Ascencion
Founder pwn4pwn.org
I would be interested in marketing your device in the Texas region. I have 30 + years in medical sales, the last 11 in sleep, sleep diagnostics, and dental sleep medicine. I have many contacts in the sleep industry including groups of ENT’s who are all about sleep. Let me know if I can help you in Texas.
Harry Power
Sleep Services North Texas
Hi can you pls give me a free trial becose I have a bad snoring and I feel bad to my self thanks
I need to see results of well designed clinical trials with a defined population to determine both short term & long term efficacy. Then I need to know the cost & maintenance of the device.
The most common reason for poor compliance for CPAP use is poor fit either for the mask &/or the settings.
Our group would be interested in a report of safety and efficacy findings of your first clinical trial. We would also give strong consideration to participating in future trials
I am an OSA sufferer who was CPAP non-compliant and currently use an oral device. I’d be interested in taking part in your clinical trial. I also work in the industry. Please view my LinkedIn profile, and would be interested in meeting with your team.
I would like to purchase a device
please advice
I would like to enroll in your US trials as I have moderate sleep apnea and cannot tolerate cpap. Thank you very much