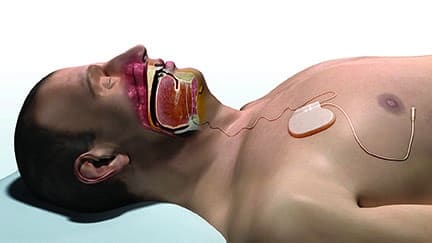
Inspire Medical Systems Inc, the manufacturer of the only FDA-approved active implantable device for obstructive sleep apnea (OSA), today announced 5-year results of the STAR clinical trial. The data were presented at the recent American Academy of Otolaryngology – Head and Neck Surgery Foundation (AAO-HNSF) annual meeting and featured in the “Best of” presentation session at the conference. The trial results demonstrated that the significant improvements derived by patients utilizing Inspire therapy were sustained following 5 years of continued use.
“The 5-year results from the STAR clinical trial are the most significant to date demonstrating the durable long-term improvement in objective and patient-reported outcomes with Inspire therapy,” says B. Tucker Woodson, MD, of Froedtert Hospital & the Medical College of Wisconsin in a release. “The investigating physicians involved in the study continue to conclude that, in CPAP intolerant sleep apnea patients who meet inclusion criteria, cranial nerve stimulation with Inspire therapy provides safe and sustained benefit.”
The presentation of the STAR 5-year results highlighted:
- Significant reduction in the severity of patients’ OSA
- Significant improvements in sleep-related quality of life
- Statistically significant reduction in snoring
- 80% of patients in the study at 5-years used the device every night
“Consistent and predictable patient outcomes are the priorities for Inspire therapy, and the foundation for continued growth in its adoption,” says Inspire president and CEO, Tim Herbert. “As such, we are excited about the growing body of clinical evidence showing Inspire therapy to be a safe and effective therapy for CPAP-intolerant patients, and are committed to expanding its use with sleep apnea patients globally.”
In another presentation at the AAO-HNSF conference, Inspire announced the launch of the ADHERE Registry to evaluate 2,500 Inspire therapy patients in the United States and Europe. Early data on 259 patients from the ADHERE Registry were presented and demonstrated positive results in patient outcomes and therapy adherence that are consistent with the STAR clinical trial.
“The ADHERE Registry is the largest collective report on the performance of Inspire therapy at leading medical centers in the world,” says Maurits Boon, MD, of the Thomas Jefferson University Hospital (TJUH) in Philadelphia. “Initial patient outcomes have been compelling to date, and we look forward to continuing to enroll patients in this important registry study.”
Finally, an independent clinical study conducted at TJUH and the University of Pittsburgh Medical Center (UPMC) entitled, “Upper Airway Stimulation for Treatment of Obstructive Sleep Apnea; An Evaluation and Comparison of Outcomes at Two Academic Centers” was published online on July 14, 2017, by the Journal of Clinical Sleep Medicine. This publication included data on 97 Inspire therapy patients which demonstrated that the treatment improves both objective and subjective patient outcomes.
The results from the publication include:
- Significant reductions in sleep apnea severity in all patients
- Significant improvements in quality of life measures in all patients
- Objective report of therapy use of over 40 hours per week
“We studied a large group of patients undergoing Inspire cranial nerve stimulation therapy,” says Colin Huntley, MD, of TJUH in Philadelphia. “We are pleased to note that the improved patient outcomes seen in the clinical setting are safe and reproducible in two separate high-volume institutions, TJUH and UPMC.”