Results from an international randomized study show that an implanted nerve stimulator significantly improves symptoms in those with central sleep apnea (CSA), without causing serious side effects.
William Abraham, MD, co-lead author and director of the Division of Cardiovascular Medicine at The Ohio State University Wexner Medical Center, presented findings from the study at the recent European Society of Cardiology Congress in Rome. The study is published by The Lancet. Unlike the more common obstructive sleep apnea, in which the airway partially collapses and causes pauses in breathing, CSA occurs when the brain fails to control breathing during sleep.
“CSA is a serious concern because it affects about a third of people with heart failure and it’s known to make the condition worse,” Abraham says in a release. “Currently, we don’t have good treatments available. Positive airway pressure devices have been used, but many patients don’t tolerate them well and a recent study showed them to be harmful.”
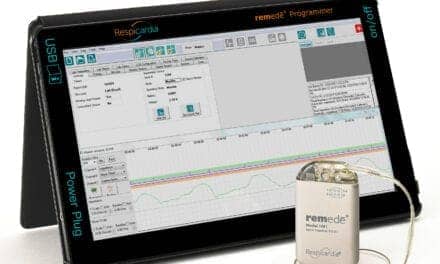
Abraham, along with lead author Maria Rosa Costanzo, MD, at Advocate Heart Institute in Naperville, Ill, led the study at 31 hospitals in the United States, Germany, and Poland. The research team tested the safety and effectiveness of a transvenous phrenic nerve stimulator made by Respicardia Inc. Much like a pacemaker, it sends a regular signal telling the diaphragm to breathe during sleep.
In the randomized study, 151 patients were implanted with the device. Ten were excluded due to non-study related medical issues or deaths, exiting the study, or missing visits. During the first six months of evaluation, 68 devices were activated for treatment, while 73 were left inactive as the control group. Between six and 12 months of follow-up, all patients received the neurostimulation treatment.
At the six month evaluation, the device reduced CSA events per hour by half or more for 35 of the 68 members (51%) of the treatment group. Only eight (11%) of those in the control group achieved the same reduction. Other important sleep measures, such as the amount of time spent with a low blood oxygen level, were also significantly improved. About a third of patients in the treatment group reported therapy-related discomfort that was resolved with some reprogramming of the device.
“Not only did we see this reduction in events per hour, the patients also rated themselves better on the Epworth Sleepiness Scale (meaning they were less sleepy during the day) and on a global assessment of their overall quality of life,” Abraham says. “This tells us the effects of neurostimulation are clinically relevant and this could be a promising therapy for those with central sleep apnea.”
In addition to Abraham, Ohio State’s Rami Khayat, MD, and Ralph Augostini, MD, participated in this research, making Ohio State one of the high enrolling centers participating in the study worldwide.
The study was funded by Respicardia. Abraham is a consultant for the company.



Have CSa am interest in the simulator are there other trials or are they in use now
I have CSA. My bipartisan is set to increase 15 then 20. It continually disturbs my sleep so I am always too tired
I would like to know how I might be a candidate to try this device.
Also, how is the bipartisan be harmful
I have CSA and CPAP options have not worked. Am I eligible for a trial?
I would appreciate any information available.
I am unable to use the CPAP equipment. I have tried on 2 occasions. I would like to know more about Inspire, including requirements for a candidate.
E. T. Parker
The device has been approved! I called the manufacturer of the pacemaker type device this week and they are sending my info to Ohio State. Thank God I live in Michigan and not terrible far away. I talked to a David, a scientist at Respicardia.
I was diagnosed in 2008 with CSA and tried using a V-Pap machine but never was able to get use to it so I stopped wearing it after trying off and on for over 1 year. Last Sept, 2017, I was diagnosed with Severe Pulmonary Hypertension. I have been diagnosed in Groups 1, 2, & 3 meaning my sleep apnea is contributing to my SPH. I would like to know if I could be a candidate for this treatment. I live in Toledo, Ohio and am receiving my treatments for Pulmonary, Cardiac, & Rheumotology all at The University of Michigan.