Australian researchers have discovered that a bedtime nasal spray has the potential to reduce the severity of obstructive sleep apnea (OSA) in people and lower their blood pressure.
The research is published in The Journal of Heart and Circulatory Physiology.
The study aimed to determine the effects of a new potassium channel blocker nasal spray on OSA severity and investigate the potential influence of different breathing approaches, such as restricted “nasal only” breathing and the physiological characteristics of those who had a favorable response.
“Potassium channel blockers are a class of drugs that block the potassium channel in the central nervous system. When used in a nasal spray, the blockers have the potential to increase the activity of the muscles that keep the upper airway open and reduce the likelihood of the throat collapsing during sleep,” says lead author Amal Osman, PhD, in a release.
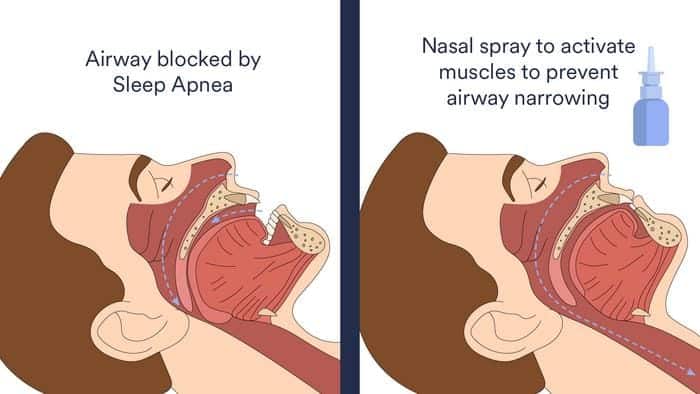
Using a randomized blind trial, 10 people with OSA were given either the potassium blocker nasal spray, a placebo nasal spray, or the potassium nasal spray in combination with restricted “nasal only” breathing.
Seven out of the 10 people responded to the potassium channel blocker nasal spray showing a reduction in the frequency of upper airway collapsing episodes during sleep and lower blood pressure the next morning. The use of the spray with restricted “nasal only” breathing did not improve quality of sleep in this trial.
“What we have discovered is that the nasal spray application of the potassium channel blocker that we tested is safe, well-tolerated. Those who had a physiological improvement in their airway function during sleep also had between 25-45% reductions in markers of their OSA severity including improved oxygen levels as well as a reduction in their blood pressure the next day,” says Osman in a release.
Professor Danny Eckert, PhD, college of medicine and public health, adds in a release, “These insights provide a potential pathway for development of new therapeutic solutions for those people with OSA who are unable to tolerate CPAP machines and/or upper airway surgery and those with a desire for alternatives to existing therapies. Right now, there are no approved drugs for treating OSA, but through these findings and future research we are getting closer to developing new and effective drugs that are safe and easy to use.”
This work was supported by an investigator-initiated research grant from Bayer.
Photo 179720947 © | Dreamstime.com



