Recent research has shown that patients treated with zaleplon report higher quality of general functioning in society than before treatment.
Insomnia comprises sleep initiation problems, but also nightly and early morning awakenings. The majority of patients suffering from insomnia are treated in an outpatient setting and continue their daily activities, including driving a car. In this context, the ideal hypnotic drug should be devoid of residual effects the morning following bedtime administration, but also after middle of the night administration.
For a long time, benzodiazepines were the first choice for pharmacological treatment to promote the relief of sleep disturbances. Although efficient in the treatment of insomnia, benzodiazepines have the disadvantage of producing next day sedation and reduced alertness. These unwanted residual drug effects influence daily activities such as on-the-job performance and driving a car. In fact, on-the-road studies have shown that driving is significantly impaired during the day following bedtime administration of flurazepam, loprazolam, flunitrazepam, and nitrazepam.1-2 The magnitude of impairing effects depends on time after intake, half-life, and administered dosage, but none of the benzodiazepine hypnotics is completely free from residual effects. Hence, usage of benzodiazepines in the treatment of insomnia is limited to bedtime administration, and patients should be warned not to drive a car.
The unfavorable drug profile of benzodiazepines led to the development of nonbenzodiazepine hypnotics such as zolpidem and zaleplon. Both agents act as agonists at the benzodiazepine GABAa receptor complex, but unlike the benzodiazepines that bind nonselectively to both type 1 and type 2 benzodiazepine GABAa receptors, zolpidem and zaleplon bind selectively to the type 1 benzodiazepine receptor. Most GABAa receptors are composed of three classes of subunits with several variants (a1-6, b1-3, g1-3). Benzodiazepine receptors can be differentiated upon their subunit structure. Type 1 receptors are composed of a1, b1-3, g2 subunits whereas type 2 receptors are a2,3,5, b1-3, g2 subunits. The functional significance of these subunits is still under investigation, but it has been established that sedation and anterograde amnesia are mediated by a1-containing receptors, whereas a2 receptors mediate anxiolytic effects.3-6 Thus, in contrast to benzodiazepines, both zaleplon and zolpidem show a high affinity and high selectivity for a1-containing receptors.
The half-life of both zolpidem (1.2-4.0 hours) and zaleplon (0.8-1.4 hours) is much shorter than those of the benzodiazepines.7 The recommended dose of both drugs is 10 mg. Taking into account their receptor specificity and short half-life, zaleplon and zolpidem were promising new drugs in the development of an “ideal hypnotic” without next-day residual effects.
Four on-the-road driving studies have been performed to determine the effects of zolpidem and zaleplon on driving ability. The on-the-road driving test was developed in the 1980s in The Netherlands and has been applied in a great number of studies investigating the effects of psychoactive drugs on driving ability in both healthy volunteers and patients. An important aspect of the on-the-road driving test is its conductance during normal traffic. The presence of normal traffic gives the on-the-road driving test a high ecological validity, which cannot be replicated in a laboratory setting or driving simulators. Hence, the driving test is a close representation of real driving.
This article will discuss the outcome of these studies and the implications for the clinical use of zaleplon and zolpidem in relation to driving safety.

Figure 1. Calculation and meaning of SDLP.
In the standardized driving test, subjects are instructed to drive a car over a 100-km (61 miles) circuit with a constant speed (95 km/hour or 58 miles/hour) and a steady lateral position within the right (slower) traffic lane. The primary parameter of the test is the standard deviation of lateral position (SDLP), that is, the weaving of the car. This is illustrated in Figure 1, (page 48).
It is evident by looking at Figure 1, that SDLP represents the amount of vehicle control. That is, higher SDLP values represent increased weaving of the car. Under normal circumstances, SDLP values of healthy volunteers with moderate driving experience (5,000 km/year) range between 18 and 22 cm (7.0 and 8.7 inches). However, highly elevated SDLP results in excursions out of lane, both into the road shoulder and/or the adjacent traffic lane. Hence, high SDLP values represent unsafe driving behavior.

Figure 2. Schematic representation of the on-the-road driving test.
The on-the-road driving test is performed using an instrumented test vehicle. As illustrated in Figure 2, a camera mounted on the roof of the car records the position of the car. The lateral position of the car is determined continuously (2 Hz) during the test. Speed is determined by the revolutions of the wheels. In the right front seat, a licensed driving instructor who is equipped with dual controls accompanies the subject. His main responsibility is to guard the subject’s safety. If the driving instructor or the subject judges that it is unsafe to continue the driving test, the test is terminated before completion.
On-the-road driving studies
The first study examining the effects of zolpidem on actual driving ability was performed in a sample of 17 women who complained of chronic sleep disturbances.8 On-the-road driving tests were performed 10 to 11 hours after administration of a single dose of zolpidem (10 mg), flunitrazepam (2 mg), and a placebo. Results from this study showed that zolpidem at 10 mg did not significantly impair driving performance after bedtime administration. In contrast, driving ability was significantly affected after flunitrazepam.
Vermeeren and associates9 compared the effects of zaleplon (10 mg and 20 mg) and zopiclone (7.5 mg) with placebo after bedtime and nightly administration. The study revealed that zaleplon (10 mg and 20 mg) did not significantly impair driving performance of 28 healthy volunteers after 6 hours (nightly administration) or 11 hours (bedtime administration). In contrast, zopiclone significantly impaired driving performance after both bedtime and night administration.
A recent study10 examining driving ability 10 hours after bedtime administration of zaleplon (10 mg) and zopiclone (7.5 mg) replicated these findings. Additionally, the same subjects performed afternoon driving tests after consuming alcohol (blood alcohol concentration <0.05%) and placebo. Driving performance after alcohol intake was significantly impaired. Zaleplon did not impair driving performance, whereas SDLP increment after zopiclone (+3.4 cm) was twice the magnitude of that observed with alcohol (+1.7 cm).
 Figure 3. Mean (+SE) of SDLP for each treatment. Ethanol (ETHA), placebo-ethanol (PLAE), placebo (PLAC), zaleplon 10 mg (ZA10), zaleplon 20 mg (ZA20), zolpidem 10 mg (ZO10) and zolpidem 20 mg (ZO20). Significant differences from placebo are indicated by asterisks (*).
Figure 3. Mean (+SE) of SDLP for each treatment. Ethanol (ETHA), placebo-ethanol (PLAE), placebo (PLAC), zaleplon 10 mg (ZA10), zaleplon 20 mg (ZA20), zolpidem 10 mg (ZO10) and zolpidem 20 mg (ZO20). Significant differences from placebo are indicated by asterisks (*).
Verster and colleagues11 examined whether residual effects of zaleplon and zolpidem affect driving ability after middle-of-the-night administration; 30 healthy volunteers participated in a seven period double-blind crossover study comparing the effects of zaleplon (10 mg and 20 mg) and zolpidem (10 mg and 20 mg) with placebo, 4 hours after middle of the night administration. For comparative purposes, the same subjects also performed afternoon driving tests after alcohol (blood alcohol concentration <0.05%) and placebo. Results from this study showed that driving performance was significantly impaired after both doses of zolpidem. Relative to placebo, SDLP increased in a dose-dependent manner after zolpidem 10 mg (+4 cm) and zolpidem 20 mg (+11 cm). Driving ability after alcohol was also significantly impaired. As illustrated in Figure 3, the magnitude of SDLP increment was significantly higher after both doses of zolpidem than after alcohol (+1 cm). In contrast, driving performance after zaleplon 10 mg (the recommended dose) and zaleplon 20 mg was not affected 4 hours after middle of the night administration.
It must be taken into account that the SDLP values shown in Figure 3 represent the average driving performance of 30 healthy volunteers; however, it is known that (adverse) drug effects can show marked differences between patients, reflected in performance on the driving test. Hence, individual driving test results (SDLP differences from placebo) for each subject are depicted in Figure 4 (page 53).
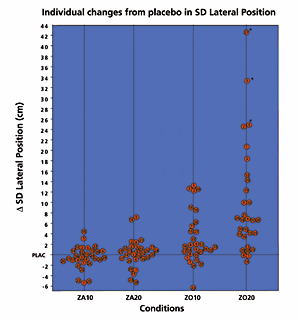 Figure 4. Individual changes in SDLP from respective placebo values (³SDLP) for zaleplon 10 mg (ZA10), zaleplon 20 mg (ZA20), zolpidem 10 mg (ZO10) and zolpidem 20 mg (ZO20). Positive values of ³SDLP indicate impairment and negative values indicate improvement of performance. Numbers in the circles indicate subjects. Subjects who did not complete their driving test are marked by asterisks (*).
Figure 4. Individual changes in SDLP from respective placebo values (³SDLP) for zaleplon 10 mg (ZA10), zaleplon 20 mg (ZA20), zolpidem 10 mg (ZO10) and zolpidem 20 mg (ZO20). Positive values of ³SDLP indicate impairment and negative values indicate improvement of performance. Numbers in the circles indicate subjects. Subjects who did not complete their driving test are marked by asterisks (*).
It is evident from Figure 4 that driving performance after zaleplon 10 mg and 20 mg matched that of placebo. In contrast, after zolpidem, SDLP changes from placebo were pronounced. Especially after zolpidem 20 mg (twice the recommended dose), driving performance was seriously impaired; the driving tests of three subjects (indicated by an asterisk) were terminated before completion due to very unsafe driving behavior.
Discussion
Epidemiological evidence12 shows that most patients do not experience sleep disturbances on a daily basis. Further, problems occur at different times during the night. Most prominent complaints accompanying chronic insomnia included middle of the night awakening (67%), difficulty returning to sleep after awakening (57%), and sleep initiation problems (56%). Thus, the need of a flexible dosing regimen for patients suffering from middle of the night or early morning awakenings requires hypnotics that act fast and are eliminated rapidly. In addition, 72% of these patients reported waking up drowsy as a major problem. Benzodiazepine hypnotics can aggravate this drowsiness and reduce alertness during the day, making insomnia a 24-hour problem. This is illustrated by the results from several on-the-road studies showing that driving performance was significantly impaired up to 17 hours after benzodiazepine intake.1,2 It is understandable that under these conditions, patients are limited in their daily functioning and are more vulnerable to developing secondary pathologies such as anxiety, depression, and mood changes. Therefore, an important aim of drug development should be to design hypnotics without residual daytime effects.
Nonbenzodiazepine hypnotics such as zolpidem and zaleplon were developed to overcome the problems experienced with benzodiazepines. On-the-road studies indicate that driving performance was not impaired the morning following bedtime administration of zolpidem (10 mg) and zaleplon (10 mg and 20 mg). Relative to the benzodiazepines, this is a relevant step forwards in the treatment of insomnia.
Regarding the use of hypnotics during the night, Vermeeren and colleagues9,10 showed that zaleplon (10 mg and 20 mg) did not impair driving ability 6 hours after administration; however, the most convincing evidence that driving is safe after using zaleplon during the night comes from the study of Verster and colleagues.11 As little as 4 hours after middle-of-the night administration, driving performance after zaleplon (10 mg and 20 mg) matched that of placebo. In contrast, zolpidem (10 mg and 20 mg) significantly impaired driving ability in a dose-dependent manner. In line, laboratory studies using a battery of cognitive and psychomotor tests showed that zaleplon did not produce significant performance impairment when administered within few hours before awakening, whereas zolpidem produced significant impairment on most tests.13-15
Conclusion
The advantage of zaleplon relative to the other hypnotics must be taken into account when prescribing a hypnotic drug to outpatients, since most of them presumably drive a car. In this context, recent research has shown that patients treated with zaleplon are more satisfied and report higher quality of general functioning than before treatment.16 This is in contrast to patients treated with benzodiazepine hypnotics who often reported reduced morning alertness and “drugged feelings.”17 The favorable adverse effect profile of zaleplon presumably enhances treatment compliance since zaleplon meets the needs of patients who want to participate fully in society.
Joris C. Verster, PhD, and Edmund R. Volkerts, PhD, are research associates at the Utrecht Institute for Pharmaceutical Sciences, Department of Psychopharma-cology, University of Utrecht, The Netherlands.
References
1. O’Hanlon JF, Volkerts ER. Hypnotics and actual driving ability. Acta Psychiatr Scand. 1986;74:S95-S104.
2. Van Laar MW, Volkerts ER. Driving and benzodiazepine use. Evidence that they do not mix. CNS Drugs. 1998;10:383-396.
3. Sanger DJ, Benavides J, Perrault G, et al. Recent developments in the behavioral pharmacology of benzodiazepine (w) receptors: evidence for the functional significance of receptor subtypes. Neurosci Biobehav Rev. 1994;18:355-372.
4. Doble A. New insights into the mechanisms of action hypnotics. J Psychopharmacol 1999;13:S11-S20.
5. Dämgen K, Lüddens H. Zaleplon displays a selectivity to recombinant GABAA receptors different from zolpidem, zopiclone, and benzodiazepines. Neurosci Res Commun. 1999;25:139-148.
6. Rudolph U, Crestani F, Möhler H. GABAa receptor subtypes: dissecting their pharmacological functions. Trends Pharmacol Sci. 2001;22:188-194.
7. Greenblatt DJ, Harmatz JS, Von Moltke, et al. Comparative kinetics and dynamics of zaleplon, zolpidem, and placebo. Clin Pharmacol Ther. 1998;64:553-561.
8. Vermeeren A, O’Hanlon JF, DeClerck AC, et al. Acute effects of zolpidem and flunitrazepam on sleep, memory and driving performance, compared to those of partial sleep deprivation and placebo. Acta Therapeutica. 1995;21:47-64.
9. Vermeeren A, Danjou PE, O’Hanlon JF. Residual effects of evening and middle-of-the-night administration of zaleplon 10 and 20 mg on memory and actual driving performance. Hum Psychopharmacol Clin Exp. 1998;13:S98-S107.
10. Vermeeren A, Riedel WJ, van Boxtel MPJ, et al. Differential residual effects of zaleplon and zopiclone on actual driving: a comparison with a low dose of ethanol. Sleep. 2002;25:224-231.
11. Verster JC, Volkerts ER, Schreuder AHCML, et al. Residual effects of middle-of-the-night administration of zaleplon and zolpidem on driving ability, memory functions, and psychomotor performance. J Clin Psychopharmacol. 2002;22:576-583.
12. Ancoli-Israel S, Roth T. Characteristics of insomnia in the United States: results of the 1991 National Sleep Foundation survey. I. Sleep. 1999;22:S347-S353.
13. Danjou P, Paty I, Fruncillo R, et al. A comparison of the residual effects of zaleplon and zolpidem following administration 5 to 2 hours before awakening. Br J Clin Pharmacol. 1999;48:367-374.
14. Troy SM, Lucky I, Unruh MA, et al. Comparison of the effects of zaleplon, zolpidem, and triazolam on memory, learning, and psychomotor performance. J Clin Psychopharmacol. 2000;20:328-337.
15. Hindmarch I, Patat A, Stanley N, Paty I, Rigney U. Residual effects of zaleplon and zolpidem following middle of the night administration five hours to one hour before awakening. Hum Psychopharmacol. 2001;16:159-167.
16. Wan G, Roth T, Levine P, Panish J, Cantillon M. General functioning and satisfaction in patients using zaleplon. Int J Neuropsychopharmacol. 2002;5:P1. W.030,S81.
17. Wan G, Levin G, Tan J, Panish J, Cantillon M. Zaleplon shows a more lifestyle flexible hypnotic in insomnia patients. Int J Neuropsychopharmacol. 2002;5:P1.W.029, S81.






