Oral appliance maker SomnoMed Ltd reports its quarterly activities for the period ended September 30 (Q1 FY23).
During the first quarter, SomnoMed passed the milestone of 740,000 patients treated worldwide. It also hosted its second annual SOM Summit, which brought together the sleep medicine practitioners from North America and globally to drive awareness and education of COAT technology for the treatment of obstructive sleep apnea (OSA).
Rest Assure, the company’s first ever in-built technology-enabled oral appliance, remains on schedule with the focus on the preparation of documentation required for regulatory submissions.
SomnoMed continues to proactively deliver on a range of initiatives to secure its supply chain with the aim of limiting any negative impact of inflation pressure on margins.
SomnoMed managing director, Neil Verdal-Austin says in a release, “The September quarter saw the company deliver financial growth in line with guidance, increase the awareness of COAT for the treatment of OSA and progress the development of our technology-enabled oral appliance, Rest Assure.
“Whilst the Northern Hemisphere summer represents a seasonally lower revenue quarter in Q1, total group revenue was up 19% year on year. Our strong sales results and growing level of patients treated validates our Medical Initiative strategy and confirms that the exceptional ‘first time fit’ quality and durability of our product range is superior and preferred by clinicians and patients world-wide for the treatment of mild to moderate OSA.”
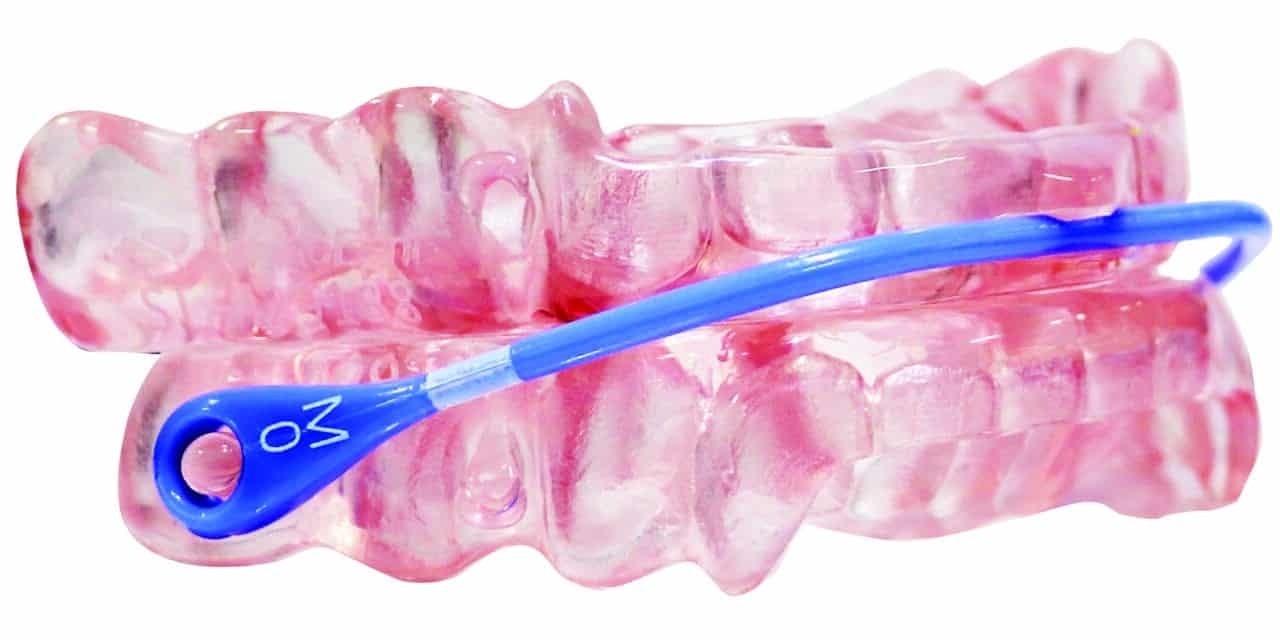
Q1 FY23 revenue of $18.5 million, up +19% (+21% in constant currency) versus previous corresponding period, which the company states reflects the growing demand for COAT technologies, AVANT (pictured) and Herbst Advance Elite, and the investment the company has made into sales teams and business development channels in all regions.
Cashflow from operations was negative $1.9 million after excluding one-off payments relating to annual license fees and bonuses. Cash investments in R&D were mainly related to the Rest Assure technology of c.$1 million. The company maintains the forecast for FY23 EBITDA1 of at least $2 million and a CAPEX investment of c.$7 million, which includes expected technology innovation spend of c.$3 million.
SomnoMed remains funded to execute on its growth priorities with available cash of $12 million as at September 30, and a further $6 million of the new debt funding arrangement to be drawn down towards the end of Q2 FY23.
Business conditions continued to improve across SomnoMed’s key regions of North America, Europe, and Asia Pacific as the impacts of COVID-19 on the medical sector and broader dental community continued to reduce.
SomnoMed Q1 in North America
The North America market experienced another positive quarter with revenue up +44% (+33% in constant currency) versus previous corresponding period, driven by increased investment in sales and marketing efforts in the region driving demand for the product range, especially the AVANT and Herbst Advance Elite.
The direct impact of the Medical Initiative continues to validate the company’s strategic direction. The additions made to both the business development team and account management personnel indicate the depth and strength of the company’s integrity and value-based sales methodologies across both the medical and dental communities.
Q1 in Europe
Revenue for the quarter was up +4% (+12% in constant currency) versus the previous corresponding period. Patient demand for the company’s COAT technology remains strong across core countries within Europe, driven by strong positive reimbursement trends and a growing acceptance of the benefits of oral appliances for mild and moderate OSA patients.
An investment was made in sales training across the region involving a strategic methodology approach to solution selling.
Q1 Asia Pacific
Asia Pacific quarterly revenues were up +31% (+33% in constant currency) versus the previous corresponding period. This region has returned to deliver impressive growth for the quarter while continuing with its clinical education program and the investment in new sales and marketing resources to advance the adoption of oral appliances within the medical sector.
Rest Assure Smart Oral Appliance Development Status
SomnoMed introduced Rest Assure, its first ever in-built technology-enabled oral appliance, in February 2022. Rest Assure is designed to address the lack of overnight monitoring and objective data in oral appliances.
The second patient validation study has been completed and the results are currently being analyzed by the principal investigators. The Rest Assure project remains on schedule, with SomnoMed’s focus now on the preparation of documentation required for regulatory submissions to the FDA (USA), TGA (Australia), and for CE marking (Europe). Rest Assure will be commercialized once these approvals are received.
SOM Summit 2022
During the first quarter, SomnoMed hosted the second annual SOM Summit, a conference that the company states is a key pillar in the awareness and education process for delivering COAT for the treatment of OSA, allowing SomnoMed to further develop relationships within the medical field that will build on the Medical Initiative strategy and to “treat more patients more effectively.”