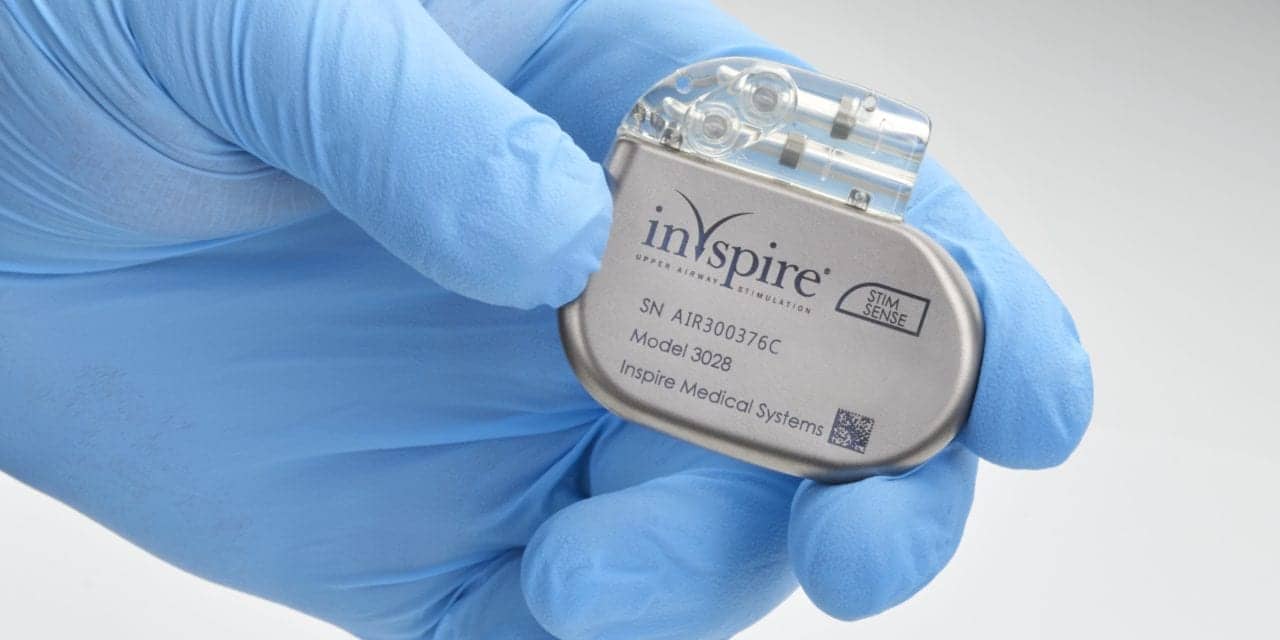
The US Food and Drug Administration (FDA) has approved an improved surgical implant procedure for Inspire Upper Airway Therapy for obstructive sleep apnea. The newly approved procedure eliminates one incision with a revised placement of the pressure sensing lead—and reduce the average procedure time for Inspire therapy by approximately 20%.
“Delivering consistent positive patient outcomes remains Inspire’s number-one priority, and the receipt of FDA approval for this two-incision procedure represents further evidence of our commitment to the continuous improvement of Inspire therapy,” says Tim Herbert, president and CEO of Inspire Medical Systems, in a release. “The new procedure places the sensor in the intercostal muscles directly behind the neurostimulator pocket, thereby eliminating the need for a third incision lower on the chest and the additional requirement to tunnel the sensing lead under the skin to the neurostimulator. A number of ENT surgeons highly experienced with the current Inspire implant procedure initiated the development of this new surgical technique. With this approval, we now intend to leverage our broad network of leading ENT’s to further improve the Inspire procedure and enhance therapy outcomes.”
Eugine Chio, MD, director of the sleep surgery program at the Ohio State University Wexner Medical Center, says in a release, “Placement of the sensor in this revised location simplifies the overall procedure, while maintaining the strong safety and efficacy profile of the therapy. In partnership with several of my colleagues, we conducted a clinical study comparing this new two-incision approach to the initially approved three-incision procedure. The results showed no difference in the safety or effectiveness of the therapy, but yielded a statistically significant decrease in surgical time to just under 100 minutes on average, which is a 26-minute reduction from the three-incision procedure time.”
The elimination of the third incision, as well as the required lead tunneling procedure, is expected to minimize the risk of breast and chest wall injury, reduce operative time, and decrease post-operative pain. Training for the two-incision procedure will begin immediately.