RBD expert Carlos H. Schenck, MD, starts at the beginning.
In 1982, Carlos H. Schenck, MD, evaluated his first sleep medicine patients. A staff psychiatrist at Hennepin County Medical Center in Minneapolis, Schenck had long held an interest in neurophysiology. So it was serendipitous timing that the Minnesota Regional Sleep Disorders Center (MRSDC)—which had opened its doors at Hennepin County Medical Center in 1978 as a department of neurology initiative—was expanding its physician base to become a multidisciplinary sleep center. Doctors in other departments were asked if they were interested in practicing some sleep medicine. Since much of sleep medicine and sleep science involves neurophysiology during sleep, Schenck signed up.
As it happened, Schenck’s first day working part-time at the sleep center coincided with the appointment of a patient named Donald Dorff. Dorff told Schenck about his violent dreams and how he’d wake up injured and with his bedroom in disarray, apparently the result of unknowingly acting out those dreams. Dorff was scheduled for an overnight sleep study.
During the sleep study, Schenck witnessed Dorff’s thrashes, twitches, jerks, kicks, and punches—all occurring while Dorff was sound asleep. “It was exciting for me,” Schenck recalls. Equally exciting was the conversation between sleep center director Mark W. Mahowald, MD, (now retired but still making regular appearances at the MRSDC) and sleep lab manager/chief technologist Andrea Patterson, RPSGT, REEGT, (now deceased) the next morning. Had Dorff’s thrashes, twitches, jerks, kicks, and punches occurred during REM or non-REM sleep? Were they seizures? What disorder was this? Eventually they all agreed: The dream-enacting activity could only have occurred during REM sleep.
Patient Dorff is now recognized as the index case for REM sleep behavior disorder (RBD), a parasomnia of abnormal behavioral release during rapid eye movement (REM) sleep and characterized by the loss of REM atonia, that is, the loss of the muscle paralysis that is supposed to accompany REM sleep.1
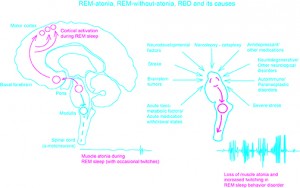
Schematic diagram. Left: The pons, the site for generating REM sleep, simultaneously sends ascending activating signals (in red) to the motor cortex and descending inhibitory signals (in blue) to the spinal cord alpha-motoneurons via the medulla, to result in REM atonia, with brief, benign twitches in REM sleep. Right: A range of clinical insults can cause REM without atonia, increased twitching in REM sleep, and RBD, with disinhibition of the REM atonia pathway indicated by the red color replacing the blue color on the left side. [The specific neuronal groups and pathways underlying this schematic diagram are contained in the basic science chapters of a new book published by Springer.] Original art courtesy of I.E. Wong Fong Sang, MSc biomedical sciences/neurosciences; PhD candidate in neurobiology (expected completion in March 2019), Johannes Gutenberg University, Mainz, Germany. Reprinted with permission from I.E. Wong Fong Sang.
The book got its start in March 2015 in Seoul, South Korea. Associate editors Birgit Högl, MD, and Aleksandar Videnovic, MD, were fresh off serving as editors of a different Springer-published book on sleep. At the World Association of Sleep Medicine congress, they approached Schenck at an early morning meeting about working together on a book about RBD. Schenck jokes that they took advantage of his jet lag so he could only say yes, but in a more serious tone, he adds that he has no regrets about embarking on the project, since it was to become the first textbook on RBD.
Schenck wrote or cowrote 10 chapters, and selected experts—many of whom are part of the international RBD study group—to write additional chapters on their fields of RBD research expertise.
The book is particularly timely. An epidemiologic study recently found the prevalence of polysomnography-confirmed REM sleep behavior disorder to be more than 1% in the general population. “That’s the same prevalence as schizophrenia,” Schenck says. “Most people know about schizophrenia. More people need to know about RBD.” What’s more, the study observed an equal male-female ratio of RBD in the general population.2 Because RBD predicts future parkinsonism, this means women—who typically have less aggressive symptoms—must be diagnosed, particularly if neuroprotective therapies become available to prevent the later development of parkinsonism and its associated dementia.
The book was unveiled at a “Tuesday Noon Meeting” at the MRSDC, a vital weekly focal point for discussing new and challenging patients, new ideas, new findings, and more. Soon after the unveiling, Schenck spoke with Sleep Review about the clinical implications of RBD diagnosis.

Neurologist Mark W. Mahowald, MD, who has an EEG expertise background, points to a PSG tracing, as Schenck and Ranji Varghese, MD, look on. Mahowald was sleep center director at the time the index case of RBD was identified. Varghese, trained in sleep medicine at the Mayo Clinic, is the current director of the MRSDC.
Sleep Review (SR): Other sleep disorders can mimic RBD symptoms. For example, “OSA pseudo-RBD” occurs when patients with severe obstructive sleep apnea are misdiagnosed with REM sleep behavior disorder due to aggressive motor-behavioral events with dreaming that can happen during apnea-triggered arousals. Can you speak about the differential diagnosis? Why is it important to get an RBD diagnosis correct?
Schenck: First, it is important to make the correct diagnosis of dream-enacting behaviors so proper therapy can be initiated to control the underlying disorder causing the behaviors and prevent injury to the affected person and bed partner. However, to make the correct diagnosis, the sleep physician and sleep laboratory should have experience with evaluating parasomnia patients both clinically and in the sleep laboratory with a sufficient PSG [polysomnography] montage and simultaneous time-synchronized video recording, as described in the book.
Peer-reviewed journal articles have identified the following conditions, besides RBD, that can cause aggressive dream-enacting behaviors: OSA pseudo-RBD (usually severe OSA); non-REM sleep parasomnias (sleepwalking, sleep terrors); nocturnal seizures; and most recently periodic limb movement (PLM) disorder pseudo-RBD (with very elevated PLM indices). In these reports, the treatment of the underlying disorder controlled both the underlying disorder and the dream-enacting behaviors. Also, REM atonia was preserved during both the initial and follow-up vPSG [videoPSG] studies, thereby excluding RBD as the basis for dream-enactment.
Furthermore, sleep clinicians need to be aware of the diagnostic entity of parasomnia overlap disorder, a combination of RBD and non-REM parasomnia that the MRSDC first identified in 1997 with a series of 33 cases,3 and which is recognized in the International Classification of Sleep Disorders-Third Edition (ICSD-3). These patients can have dream-enactment with RBD and with their non-REM sleep parasomnia. Finally, part of the comprehensive evaluation of dream-enactment is obtaining a careful medication history, since most antidepressants (except for bupropion) and other medications (eg, beta-blockers) can trigger RBD.
Second, the importance of diagnosing RBD correctly, with PSG confirmation, and excluding other disorders, is because we now know that more than 80% of RBD patients age 50 years and older will develop a parkinsonian disorder, on average 10-15 years from the time they develop RBD.4 The implications of this finding will be discussed more during this interview.
SR: We typically think of most patients with RBD as being middle-aged or old men. What about women and RBD?
Schenck: Women can certainly have RBD, but it is generally milder and with fewer injuries. So they present for medical attention much less frequently than men, who have more aggressive and injurious dream-enacting behaviors.
However, it is the presence of RBD, with the loss of REM atonia, and not the severity of RBD, that is the indicator of future parkinsonism. This means that once disease-modifying therapies are identified to slow down or halt the progression from “idiopathic RBD” to parkinsonism, then the women with mild RBD (and also the men with mild RBD) must be found so they can also receive the protective therapy.
In this scenario, sleep medicine clinicians should alert primary care and geriatric physicians and nurses about RBD and the risk of parkinsonism and dementia and encourage the use of simple RBD screening questionnaires that can be administered to older patients and their spouses.
SR: Can young people have RBD?
Schenck: People of all ages can have RBD, beginning in childhood when RBD is most often associated with narcolepsy, but also with the parasomnia overlap disorder, and more rarely brainstem tumors and other neurological conditions. The treatment of cataplexy or clinical depression in childhood with antidepressant medications can also trigger RBD. This is true for adults as well.
SR: Can animals have RBD? Have we learned anything from animal models?
Schenck: Dogs and cats have been reported in the veterinary medicine literature to have RBD with injuries, prompting their owners to bring their pets to the animal clinic for evaluation and treatment. What is so interesting and gratifying is that experimental animal models of RBD in cats, rats, and mice have been developed since 1965, which have provided key insights into the brainstem nuclei and circuits responsible for REM atonia—the targets for the parkinsonian disease process—and for understanding RBD in humans. Now the therapies developed for treating RBD in humans are being used by veterinarians to treat naturally occurring RBD in animals. This is the completion of a special human-animal therapeutic circle.
SR: How much loss of muscle paralysis during REM sleep must be observed for you to be confident in making a diagnosis of RBD?
Schenck: This is one of the most common questions asked after my lectures on RBD. The general consensus in the RBD research community, based on carefully gathered data reported in peer-reviewed journal articles, is that about a quarter of REM sleep should have loss of REM atonia. However, this is not a rigid cut-off. The ICSD-3 diagnostic criteria for RBD even allow for the diagnosis if there is modest loss of REM atonia, but if an episode of dream-enacting behavior was recorded during REM sleep and there is a convincing clinical history of RBD.
SR: RBD has been linked to the development of Parkinson’s disease later in life. Several studies found that for people who develop RBD after age 50, there is more than an 80% chance of conversion to Parkinson’s disease or its related dementia.4 How do you break that news to a patient?
Schenck: This is a very important topic that is carefully dealt with in chapters 22 and 23 of the RBD textbook. For patients age 50 years and older, there needs to be some mention about the future risk of conversion to parkinsonism, if for no other reason than the presence of this high risk on the internet, and so either the patient, the spouse, other family, or friends will find out. Just Google “RBD” and the Parkinson’s connection pops right up.
So it is clearly far better for the patient to hear this news first from the doctor than from someone else. The thorny issue is exactly how to break the news, and here is where the art of practicing medicine comes into play. The educational level and extent of medical sophistication on the part of the patient are important factors, along with the personality style (eg, obsessional, detail-oriented versus impressionistic, casual style).
The most simple and basic information to impart to a particular patient would be that “because of your RBD, there is a risk of your developing a brain disorder at some time in the future, and you should follow up regularly with your doctor and discuss this matter with him or her.” Then the doctor can field follow-up questions from the patient and spouse and proceed from there. On the other hand, for detail-oriented sophisticated patients, full disclosure of the risk for future parkinsonism should take place. There is a positive side to this scenario: many RBD patients are interested in being enrolled in future neuroprotective studies, and they are also willing to be enrolled in studies on biomarkers to determine which cluster of biomarkers in “idiopathic” RBD can best predict phenoconversion within 2-5 years, which makes research funding feasible. The RBD textbook covers all these issues in various chapters.
SR: What about patients who are younger at the time of diagnosis? Will they also convert to Parkinson’s?
Schenck: This is another common question I am asked. The answer is twofold.
First, we don’t have the data yet on the longitudinal outcome for RBD patients under the age of 50 years, so currently we don’t know.
Second, there are retrospective reports showing how presumed RBD (namely, dream-enactment but without vPSG confirmation) began decades (such as in late adolescence or early adulthood) before the emergence of parkinsonism. So the younger RBD patients should be told they may carry a risk, but that we are not sure. They should follow up with their doctor, who can provide updated information based on new research findings. We also don’t know the natural history of parasomnia overlap disorder and whether it carries a risk for parkinsonism.

Varghese (right), with Schenck, holds a 256-electrode high density (HD) EEG head net, which is a tool employed by the MRSDC in a collaboration with the University of Wisconsin-Madison. They monitor non-REM parasomnia patients (who have sleepwalking, sleep terrors, sleep related eating, etc) to capture behavioral events in the sleep lab, and then correlate the behaviors with the HD-EEG findings.
SR: What is the status of the scientific research that is trying to figure which RBD patients will convert to Parkinson’s? And at what biomarkers are they looking?
Schenck: This is an area of intense scientific research, for the reason we discussed earlier (in regard to identifying the highest-risk RBD patients for converting to parkinsonism within 2-5 years, so they can be enrolled in neuroprotective trials). Chapter 36 by Ron Postuma, MD, MSc, addresses the topic of biomarkers, with some of the most important ones being loss of smell, vision changes, subtle motor abnormalities (including gait), EEG slowing, abnormal brain imaging findings, mild cognitive impairment, constipation, depression, etc.
I should point out that a number of biomarkers in isolation are quite nonspecific, such as constipation or depression. But when they are clustered together with other biomarkers, especially in the powerful context of RBD, then they can become predictors of imminent phenoconversion.
SR: What is the status of therapies for RBD? Are any therapies being investigated that may prevent the later development of Parkinson’s?
Schenck: Clonazepam and melatonin taken at bedtime are the usually effective firstline therapies of RBD, as confirmed by an American Academy of Sleep Medicine standards of practice publication in the Journal of Clinical Sleep Medicine.5
However, an exciting publication from earlier this year in the Archives of Physical Medicine and Rehabilitation described another (complementary) therapeutic approach. Based on the correct logic that since virtually all middle-aged and older RBD patients are in the process of developing parkinsonism, the authors posit that the physical and occupational therapy that is employed in parkinsonian patients should also be started in newly diagnosed RBD patients. The aim is to maximize the extent and duration of functionality. The areas of physical therapy identified were postural muscles, trunk range of motion, movements practice (involving amplitude and speed), balance training (for fall prevention), agility training (to maintain mobility under challenging environmental and dual task conditions), aerobic exercise, and gait assessment. The areas of occupational therapy identified were securing the safety of the sleeping environment, developing compensatory measures for autonomic dysfunctions, rehabilitating cognitive dysfunction (such as memory dysfunction), and voice and speech therapy.6 The latter is to address the four components of speech that can be affected in early parkinsonism (aperiodicity, alternating motion irregularities, articulatory decay, dysfluency).
Finally, basic scientists and clinical investigators are actively searching for promising neuroprotective agents to test that may slow down or halt the progression from RBD to parkinsonism.

Schenck dedicated the first RBD textbook to Mahowald (left) and to the late Michel Jouvet, MD, PhD, pioneer in the first experimental animal model of RBD.
SR: What is the status of figuring out the cause(s) of RBD?
Schenck: Besides parkinsonism and antidepressant medications, there is a broad spectrum of neurological disorders that can cause RBD by their impact on REM atonia, as discussed in detail in the RBD textbook. Also, autoimmune diseases, paraneoplastic disorders (related to remote malignancies), and toxic-metabolic disorders can cause RBD. And a new frontier of acute stress-induced RBD needs further research. Trauma-associated sleep disorder (TSD) has been reported in a series of cases that involved some loss of REM atonia with aggressive dream-enacting behaviors in PTSD patients. Table 1 in the textbook preface lists the 46 areas of research that intersect with RBD, indicating how RBD is at the center of many important areas of clinical neuroscience and sleep medicine.
Sree Roy is chief editor of Sleep Review.
References
1. Schenck CH, Bundlie SR, Ettinger MG, Mahowald MW. Chronic behavioral disorders of human REM sleep: a new category of parasomnia. Sleep. 1986; 9(2):293-308.
2. Haba-Rubio J, Frauscher B, Marques-Vidal P, et al. Prevalence and determinants of rapid eye movement sleep behavior disorder in the general population. Sleep. 41(2);1 February 2018:zsx197.
3. Schenck CH, Boyd JL, Mahowald MW. A parasomnia overlap disorder involving sleepwalking, sleep terrors, and REM sleep behavior disorder in 33 polysomnographically confirmed cases. Sleep. 1997;20(11):972-81.
4. Schenck CH, Boeve BF, Mahowald MW. Delayed emergence of a parkinsonian disorder or dementia in 81% of older men initially diagnosed with idiopathic rapid eye movement sleep behavior disorder: a 16-year update on a previously reported series. Sleep Med. 2013;14(8):744–8.
5. Aurora RN, Zak RS, Maganti RK, et al. Best practice guide for the treatment of REM sleep behavior disorder (RBD). J Clin Sleep Med. 2010 Feb 15;6(1):85-95.
6. Johnson BP, Westlake KP. Link between Parkinson disease and rapid eye movement sleep behavior disorder with dream enactment: possible implications for early rehabilitation. Arch Phys Med Rehabil. 2018;99(2):411-5.
Featured image: Carlos H. Schenck, MD (front row, second from left), of the Minnesota Regional Sleep Disorders Center (MRSDC), shares his and other experts’ knowledge in a new textbook entitled Rapid-Eye-Movement Sleep Behavior Disorder. He unveiled the book to colleagues at MRSDC’s weekly “Tuesday Noon Meeting.” Front row (left to right): Ranji Varghese, MD, Carlos H. Schenck, MD, Muna Irfan, MD, Mark W. Mahowald, MD, Michel A. Cramer Bornemann, MD. Back row (left to right): Pava Cabaravdic, RPSGT, Erin Golden, MD, Tangy Michnowski, RPSGT, Michelle Gerow-Ellis, APRN, CNP, Cindy Farr, Constance Ullevig, RN, Mark Gehrt, RPSGT, Eduardo A. Colón, MD (chief of psychiatry), Samantha Lee Anders, PhD, LP.






My doctor, Mark Mahowald, was wonderful. He helped me and helped figure me out (not an easy thing to do). I still appreciate him.
Dr says i have rem disorder take 2mg of clonazepam is there other treatments