A recent study suggests that treatment of OSA with auto-nCPAP initiated at a patient’s home is effective and reliable.
Nasal continuous positive airway pressure (nCPAP), which was introduced in 1981 by Sullivan et al,1 has considerably improved the treatment and prognosis of patients with obstructive sleep apnea (OSA). In practice, the optimal nCPAP level is a trade-off between pressure-related side effects and efficacy in preventing upper airway obstruction during sleep. This optimal pressure level is generally determined by manual titration during an overnight polysomnography study at the sleep laboratory; however, this conventional approach is expensive, time-consuming, and labor-intensive. As a result, sleep laboratories have long waiting lists for overnight polysomnography studies, which delay the initiation of effective treatment in patients with OSA. This is of concern because untreated OSA increases cardiovascular morbidity and the risk of traffic accidents.2 Automatic nCPAP (auto-nCPAP) devices that continuously adapt the positive pressure in response to the patient’s needs3 have been developed recently and found to be at least as effective as constant-pressure CPAP4-6 in reducing sleep-disordered breathing (SDB). Because this device adjusts the pressure automatically, a titrating polysomnography study is theoretically unnecessary, and the treatment can be initiated at the patient’s home. This can be expected to save money and to shorten the time from diagnosis to use of nCPAP, as compared with conventional nCPAP. This article will discuss the outcome of a recently published multicenter randomized study conducted in Paris, comparing the efficacy and cost of conventional nCPAP initiated during a titrating polysomnography at a sleep laboratory and auto-nCPAP initiated at a patient’s home.7
Study Design
At four French sleep laboratories 35 patients were recruited based on severe obstructive OSA with an apnea-hypopnea index (AHI) Ž30 events/hour and obstructive events >80% of total events and clinical indication for nCPAP according to American Thoracic Society recommendations. None of these patients had a history of nCPAP therapy or surgery for snoring. The patients were randomized to conventional nCPAP initiated at the sleep laboratory with a titrating polysomnography (n = 17) or auto-nCPAP initiated at home (n = 18). There were no significant differences between these two groups regarding age (54.2±10.7 vs 54.3±10.9 years, respectively; NS), body mass index (BMI) (30.9±6.1 vs 33.6±6.7 kg/m2, respectively; NS), gender distribution (five women and 12 men vs 18 women and 14 men), or AHI (60.1±19.0 vs 56.2±16.1 h-1).
The auto device was used in constant mode in the conventional group and in automatic mode in the home group. In the conventional group, nCPAP pressure was titrated manually starting at 4 cm H2O during an overnight polysomnography study at the sleep laboratory. The effective pressure was the pressure at which obstructive respiratory events, including snoring and airflow limitation, disappeared in all sleep stages and body positions. Auto-nCPAP therapy was started at home by a nurse who provided instruction on use of the device. The pressure was allowed to increase from 6 cm H2O initially to no more than 16 cm H2O, based on snoring detection only, as previously described.8-9 Data recorded by the CPAP device microprocessor (pressure levels and number of hours of actual nCPAP use) were transmitted daily to the sleep physician at the sleep laboratory via a modem. After 1 week and at least 15 hours of nCPAP, the maximum pressure level delivered by the nCPAP device was determined, and the pressure range was set at 2 cm H2O above to 4 cm H2O below this level. Patients were instructed to contact the sleep physician if needed, and patients with poor tolerance were invited to switch to the other treatment.
After 2 months of nCPAP, the patients completed the Epworth Sleepiness Scale (ESS)10 and a tolerance questionnaire as previously described.6 Data on duration of nCPAP use were downloaded from the device, and nCPAP efficacy was evaluated by overnight polysomnography at the sleep laboratory. The time from study inclusion to nCPAP was calculated. Total cost of the 2-month treatment was calculated by summing the costs of hospital care; materials including nasal mask(s), mask strip(s), inspiratory circuit, expiratory valve, humidifier (but excluding the cost of the nCPAP device, which was identical in the two groups); telecommunication (phone calls and modem); and home visit(s) by the nurse (hourly cost of the nurse and transportation cost).
Results
The mean time from diagnosis to nCPAP therapy initiation was significantly longer in the group treated with conventional nCPAP initiated at the sleep laboratory than in the group treated with auto-nCPAP initiated at home (47.2±46.5 vs 11.8±15.5 days, respectively; P<0.01). This difference was ascribable to the long waiting time before the polysomnography study needed to initiate conventional nCPAP. The mean time from diagnosis to final auto-nCPAP adjustment in the 16 patients who agreed to complete the study was about 16.3±5.0 days and remained significantly lower (P<0.01) than the mean time from diagnosis to conventional nCPAP therapy initiation.
Tolerance of nCPAP
Thirty patients completed the study. The five patients who were discontinued from the study (three in the conventional group and two in the auto-nCPAP group) were unable to tolerate nCPAP therapy, which they used for less than 1 hour per night, and refused to switch to the other nCPAP group. The two patients who could not tolerate auto-nCPAP discontinued this treatment before the final adjustment. One patient in the auto-nCPAP group experienced poor tolerance during the first week, agreed to switch to conventional nCPAP therapy, and completed the study. Although mean pressure was lower in the auto-nCPAP group than in the conventional nCPAP group (8.7±1.7 vs 11.7±2.5 cm H2O, respectively; P<0.001), the tolerance questionnaire scores in study completers were similar in the two groups.
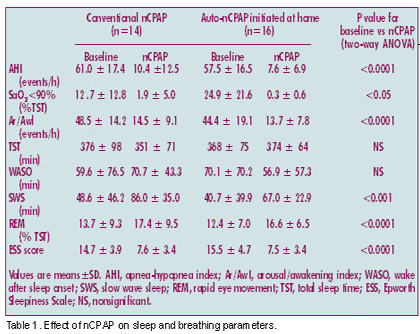
Efficacy of nCPAP Therapy
Both nCPAP modes produced comparable significant improvements in sleep respiratory parameters such as AHI and night oxygenation (Table 1). The AHI returned to normal (10 per hour of sleep) in 11 of the 14 study completers in the conventional nCPAP group. Of the three remaining patients, one had an AHI slightly above normal (11 per hour of sleep) but two had an AHI greater than 20 per hour of sleep. This inefficacy of nCPAP was ascribable in one patient to failure to determine the effective nCPAP pressure level during the overnight polysomnography; the other patient was unable to tolerate the effective nCPAP pressure, which was consequently decreased by 2 cm H2O as recommended by Sériès et al.11 In the auto-nCPAP group, AHI returned to normal in 12 of the 16 study completers; of the four remaining patients, two had an AHI value of less than 15 per hour of sleep with nCPAP, most of the remaining respiratory events being hypopneas. Sleep structure was largely restored in both groups, although the amount of slow-wave sleep remained subnormal. There was no significant difference between conventional nCPAP and auto-nCPAP regarding effects on sleep parameters. The Epworth Sleepiness Scale scores showed similar improvements in daytime sleepiness in the two groups. Finally, analysis of the questionnaires showed that all the patients but two in the conventional nCPAP group and all but one in the auto-nCPAP group were “satisfied” or “very satisfied” with their nCPAP treatment.
Compliance with nCPAP
Compliance with nCPAP as assessed by the cumulative duration of CPAP use during the 2-month study period was not significantly different in the two groups, but tended to be longer in the conventional nCPAP group (5.3±1.4 vs 4.5±1.7 hours per night with the auto-nCPAP; NS).

Cost of nCPAP Therapy
Costs of nCPAP therapy were determined in Euros per subject at the end of the 2-month study. Hospital costs were significantly lower in the auto-nCPAP group, in which all the patients but one had a single polysomnography study at the sleep laboratory (602 ± 229 vs 1,220 ± 317 Euros, respectively; P<0.001). Telecommunication costs were significantly higher in the auto-nCPAP group because of the modem transmission of nCPAP data from the home to the sleep laboratory (155 ± 7.5 vs 9 ± 5.5 Euros, respectively; P<0.001). Costs of materials and home visits by the nurse were comparable in both groups. The total cost of the 2-month treatment in Euros was significantly lower in the auto-nCPAP group than in the conventional nCPAP group (1,264 ± 352 vs 1,720 ± 455 Euros, respectively; P<0.01). Table 2 (page 30) gives an estimation of costs in US dollars, based on a recent North American study of the costs of polysomnography, a follow-up visit, and equipment for nCPAP initiation.12 In this study, the cost of polysomnography was about $1,190. We assumed that the nurse visits could be replaced by office visits ($70 per visit) and that the cost difference between the auto-CPAP device and the conventional CPAP device (including the nasal mask, headgear, and filters) would not be above the range ($1,100 to $1,420) suggested by this study.12 Although these assumptions probably led us to overestimate the cost of auto-nCPAP, we found a significant difference of about $500 in favor of auto-nCPAP.
Discussion
The aim of this study was to conduct a randomized comparison of constant (conventional) nCPAP and of automatically adjusted nCPAP (auto-nCPAP) without a titration polysomnography study in the laboratory. In earlier studies, auto-nCPAP and constant nCPAP were equally effective in correcting SDB and daytime sleepiness. Furthermore, compliance with the two treatments was similar. The present study confirmed these data and showed that auto-nCPAP used less financial and sleep laboratory resources than did constant nCPAP. In addition, auto-nCPAP reduced the time from diagnosis to treatment initiation from more than 6 weeks to 2 weeks.
Over the past few years, several strategies have been suggested to improve the cost-effectiveness of nCPAP and to shorten waiting lists in sleep laboratories. One of these strategies is a single split-night study for the diagnosis of OSA and nCPAP titration; however, Sanders et al13 found that the nCPAP pressure level determined during the split-night study had to be changed in as many as 45% of patients. In keeping with this finding, Yamashiro and Kryger14 reported that the effective nCPAP pressure level was underestimated during a split-night study in 58% of patients. Failure to determine the optimal nCPAP pressure level may result in nCPAP failure, more frequent outpatient visits for nCPAP problems, and a need for further sleep studies, thus canceling out the initial cost savings. Coppola and Lawee15 suggested an attractive approach for treating OSA, in which nCPAP pressure is adjusted over several nights at home based on reports of symptoms by both the patient and the bed partner. Ambulatory recordings of cardiorespiratory variables confirmed that the pressure thus determined was effective; however, the patients were selected, raising the possibility that this method may not be appropriate in all patients. In addition, methodological weaknesses15 may include the adjustment of nCPAP in a somewhat arbitrary fashion based on subjective reports of reduced snoring and daytime sleepiness and the absence of data on sleep architecture and arousal rates. Nevertheless, this retrospective study is the first formal study of nCPAP treatment not initiated at a sleep laboratory.
Sériès et al5 were among the first investigators to initiate auto-nCPAP at home without a titration sleep study. They demonstrated that auto-nCPAP can be used within a pressure range extending from 3 cm H2O above to 4 cm H2O below a reference pressure level calculated using a formula that includes anthropometric parameters and OSA severity indices. We used a very similar approach in our auto-nCPAP group, except that the reference pressure was the pressure required to prevent snoring. Similar to Sériès et al,5 we found that auto-nCPAP was as effective as conventional nCPAP in improving sleep quality and breathing abnormalities. Although this result should be considered with caution given the relatively small number of patients, it suggests that any difference between conventional nCPAP and auto-CPAP initiated at home is modest at most and therefore should not be taken as militating against the new method, which ensures faster treatment at lower cost. Furthermore, we observed decreases in the cost of treatment initiation and in the time from diagnosis to use of nCPAP in the auto-nCPAP group, as compared to the conventional nCPAP group. The difference in the cost of the first 2 months of treatment was about 500 Euros. We used the mean costs of polysomnography, a follow-up visit, and the equipment needed for nCPAP initiation established in a recent study12 to estimate the total cost of the 2-month treatment with both methods in North America. We found a significant difference between the two methods of about $500. More important, patients treated with auto-nCPAP required fewer polysomnography studies. Elimination of the need for a titrating polysomnography study translated into a shorter time from diagnosis to use of nCPAP in the auto-nCPAP group. Thus, more widespread use of auto-nCPAP would probably shorten polysomnography waiting lists. This is an important advantage given that OSA is a significant independent contributing factor to traffic accidents and that use of nCPAP considerably reduces the traffic accident risk in OSA patients.2 Most of our patients were drivers, and French law, as in some US states,16 prohibits patients with untreated OSA from driving; these facts underline the importance of initiating nCPAP promptly in patients with OSA.
Improving Compliance
To improve compliance with nCPAP, it is essential that the patient be able to discuss the treatment with a health care professional. The professional should explain the principles of nCPAP, emphasizing that the nCPAP device adapts to the patient, not the opposite. A wide range of masks are available, and great care should be taken in choosing the model best suited to the patient. The final objectives of nCPAP are well-being, improved quality of life, and full compliance. In our study, the patients in the auto-nCPAP group, who had no prior experience with nCPAP, received education at their home by a nurse who provided detailed explanations and demonstrated proper use of the auto-nCPAP device; however, this educational intervention may be less effective than the education received during a titration polysomnography study. This may explain the nonsignificantly poorer compliance in the auto-nCPAP group as compared with the conventional nCPAP group. Nevertheless, evidence has been reported that a simple intervention can improve compliance with nCPAP.17
The data from this study and from previous ones suggest that treatment of OSA with auto-nCPAP initiated at a patient’s home is effective and reliable. Such a simplified strategy can obviate the need for nCPAP titration, thus shortening polysomnography waiting lists, allowing prompter use of nCPAP therapy, and reducing the total cost of nCPAP therapy; however, much attention should be paid to nCPAP education provided at the patient’s home, as it appears to be of crucial importance for proper auto-nCPAP use.
Carole Planès, MD, PhD, is a sleep physician at the sleep laboratory from the Department of Physiology, Ambroise Paré Hospital (Assistance Publique des Hôpitaux de Paris), University Paris, France.
References
1. Sullivan C, Issa F, Berthon-Jones M, Eves L. Reversal of obstructive sleep apnoea by continuous positive airway pressure applied through the nares. Lancet. 1981;i:862-861.
2. McNicholas W. Sleep apnea and driving risk. Eur Respir J. 1999;13:1225-1227.
3. Berthon-Jones M. Feasibility of a self-setting CPAP machine. Sleep. 1993;16:S120-S123.
4. Meurice J-C, Marc I, Sériès F. Efficacy of auto-CPAP in the treatment of obstructive sleep apnea/hypopnea syndrome. Am J Respir Crit Care Med. 1996;153:794-798.
5. Sériès F, Marc I. Efficacy of auto-CPAP therapy that uses an estimated required pressure treatment of the obstructive sleep apnea syndrome. Ann Intern Med. 1997;127:588-595.
6. d’Ortho M, Grillier-Lanoir V, Levy P, et al. Constant versus automatic continuous positive airway pressure therapy: home evaluation. Chest. 2000;118:110-117.
7. Planès C, d’Ortho M, Foucher A, et al. Efficacy and cost of home-initiated auto-nCPAP versus conventional nCPAP. Sleep. 2003;2:156-160.
8. Lofaso F, Lorino A-M, Duizabo D, et al. Evaluation of an auto-nCPAP device based on snoring detection. Eur Respir J. 1996;9:1795-1800.
9. Berkani M, Lofaso F, Chouiad C, et al. CPAP titration by an auto-CPAP device based on snoring detection: a clinical trial and economical considerations. Eur Respir J. 1998;12:759-763.
10. Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14:540-545.
11. Sériès F, Marc I, Cormier Y, La Forge J. Required levels of nasal continuous positive airway pressure during treatment of obstructive sleep apnoea. Eur Respir J. 1994;7:1776-1781.
12. Chervin R, Muman D, Malow B, Totten V. Cost-utility of three approaches to the diagnosis of sleep apnea: polysomnography, home testing, and empirical therapy. Ann Intern Med. 1999;130:496-505.
13. Sanders M, Kern N, Costantino J, et al. Adequacy of prescribing positive airway pressure therapy by mask for sleep apnea on the basis of a partial-night trial. Am Rev Respir Dis. 1993;147:1169-1174.
14. Yamashiro Y, Kryger M. CPAP titration for sleep apnea using a split-night protocol. Chest. 1995;107:62-66.
15. Coppola M, Lawee M. Management of obstructive sleep apnea syndrome in home. Chest. 1993;104:19-25.
16. Suratt P, Findley L. Driving with sleep apnea. N Engl J Med. 1999;340:881-883.
17. Chervin RD, Theut S, Bassetti C, Aldrich MS. Compliance with nasal CPAP can be improved by simple intervention. Sleep. 1997;20:284-289.






