3B Medical Inc’s third-generation bilevel device, the Luna G3 Auto-BPAP, is available with integrated heated tubing, cellular connectivity, and 3B’s algorithm that automatically senses and adjusts pressure settings. 3bproducts.com/devices
Made of N99-rated filter material, Azimuth Sleep Solutions’ PortPatch is intended to reduce the spread of airborne particulates through the exhalation ports, anti-asphyxia valves, and carbon dioxide vents on full-face or non-rebreathing CPAP and other ventilation masks. It has FDA emergency use authorization. azimuthsleepsolutions.com
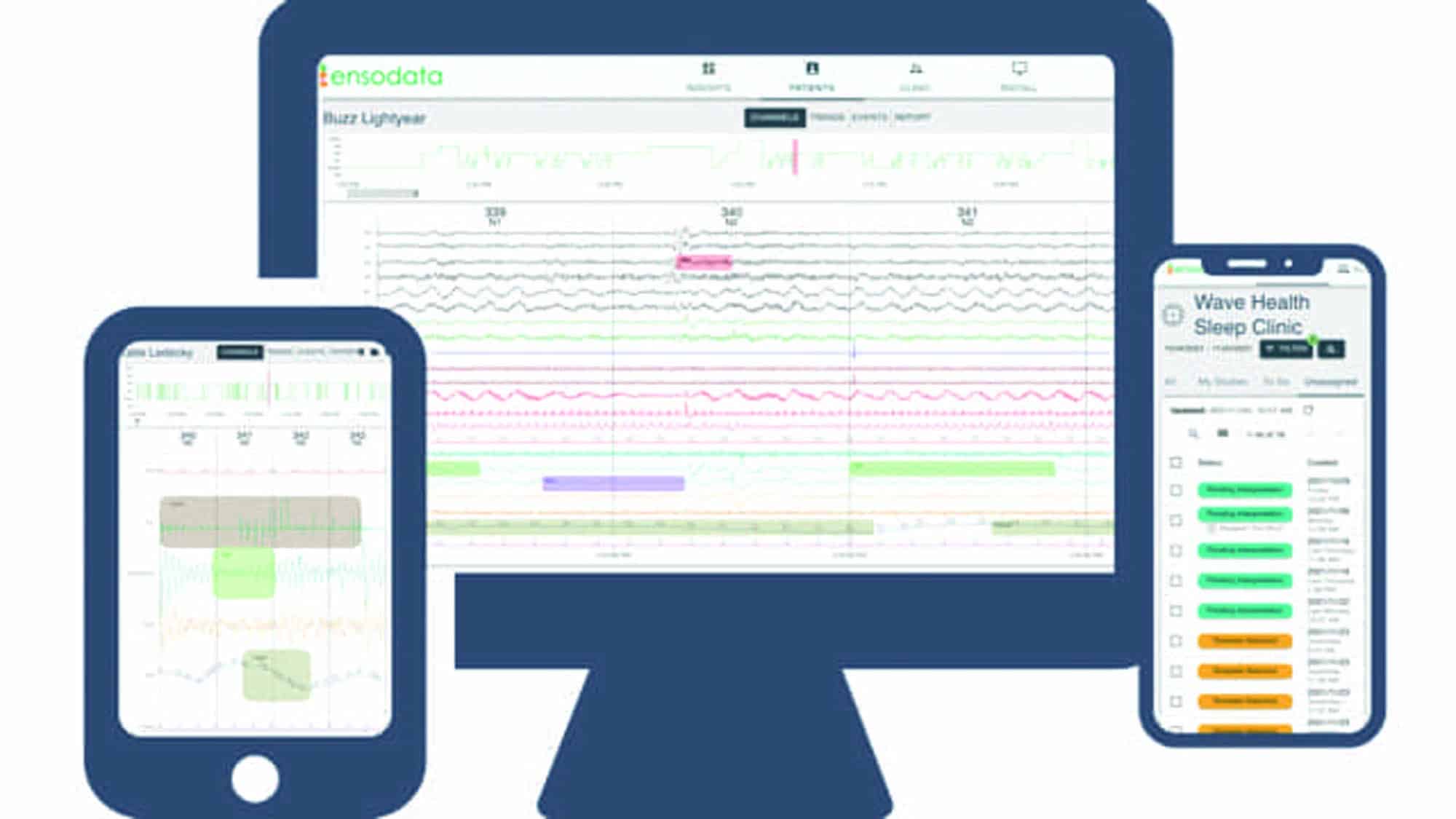
EnsoData’s artificial intelligence-assisted sleep test scoring solution, EnsoSleep, allows for review of a scored study rather than scoring from scratch. EnsoSleep is now cleared for pediatric populations ages 13 and up and includes EnsoTST, which is a total sleep time measurement for home sleep tests. ensodata.com/ensosleep
Enhancements were made to EnsoViewer, EnsoData’s FDA-cleared, interoperable, web-based, sleep study reviewing, editing, and reporting software. www.ensodata.com/ensoviewer
A newly approved procedure for Inspire Upper Airway Stimulation eliminates the need for a third incision with a revised placement of the pressure sensing lead—and reduces the average procedure time by approximately 20%. professionals.inspiresleep.com
Separately, the FDA approved a new Inspire Medical Systems Inc physician programmer platform for Inspire Upper Airway Stimulation. A new telemetry cable enables Bluetooth connectivity to the programmer tablet. It also incorporates guided workflows and interfaces with Inspire Cloud. professionals.inspiresleep.com
The FDA approved a new indication for Jazz Pharmaceuticals’ Xywav: idiopathic hypersomnia in adults. Xywav (calcium, magnesium, potassium, and sodium oxybates) oral solution was previously approved for the treatment of cataplexy or excessive daytime sleepiness in patients 7 years or older with narcolepsy. xywavhcp.com
NightWare improves sleep quality in adults with nightmare disorder or nightmares from PTSD. Using smart technology, NightWare continually evaluates the level of sleep disturbance by tracking heart rate and body movements. When a nightmare is detected, the system delivers short vibrations to interrupt it without waking the patient. nightware.com
2021 Roundup: Other New Sleep Medicine Products
These tools either did not require an FDA pathway or were approved previously but not available for purchase prior to January 2021.
3B Medical Inc launched 3B MaskFitter, a remote tool to help get patients started on the right path with mask choice and sizing, on the Apple App Store and Google Play.
AR Medical Technologies launched MaskFit AR, a mobile app powered by AI and machine learning that finds a well-fitting CPAP mask for patients.
Cadwell Industries Inc integrated EnsoData’s EnsoSleep artificial intelligence-scoring solution into Cadwell’s Easy III PSG software platform.
Itamar Medical’s WatchPAT with SleePATh application adds value by combining customizable pre/post-study and assessment questionnaires with the sleep study for a broader evaluation.
Itamar Medical also revealed WatchPAT ONE-M disposable multi-night HSAT, which enables physicians to order multi-night tests and follow-up with patients after each night of testing.
Nonin Medical Inc launched two new disposable products for its WristOx2 3150 wrist-worn pulse oximeter—a single-use wristband and a sensor.
Nox Medical launched its Nox A1s polysomnography system to the United States market. It features hospital-to-home flexibility and product enhancements to its predecessor, Nox A1.
Also, Nox Medical launched a new deep learning AI model in its Noxturnal software to detect sleep stages.
Oventus Medical introduced O2Vent telehealth therapy, a virtual treatment program including consultation, impression taking, customized oral appliance design, delivery, and follow up.
Zopec Medical LLC designed CPAP batteries that are guaranteed to last at least 8 hours, allowing for uninterruptible power supply during power outages.
Products received FDA approval or clearance between November 1, 2020 and October 31, 2021. Text based on data submitted by marketers and available on publicly accessible websites. Sleep Review makes light edits to conform with editorial style. Marketers were not charged. Sleep Review strives for accuracy in all data but cannot be held responsible for claims made by marketers. Email [email protected] to be included in the next update.
Photo 194426267 / 2021 © Nataliia Mysik | Dreamstime.com